Advertisements
Advertisements
Question
Write the chemical equations for combustion of propane.
Solution
\[\ce{\underset{\text{propane}}{C3H8(g)} + 5O2(g) ) → 3CO2(g) + 4H2O(l)}\]
APPEARS IN
RELATED QUESTIONS
Predict the possible product of the following reaction.
Chlorination of nitrobenzene
Identify the main product of the reaction:
\[\ce{H - C ≡ C - H + H - O ->[{40%} H2SO4][{1%} HgSO4]}\] _____.
Identify giving reason whether the following compound is aromatic or not.
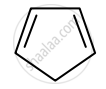
Identify giving reason whether the following compound is aromatic or not.

Name two reagents used for acylation of benzene.
Identify the compound 'Z' in the following reaction
\[\ce{C2H6O ->[Al2O3][623 K] X ->[O3] Y ->[Zn/H2O] (Z)}\]
Write the structure of the following alkanes.
5 – (2 – Ethyl butyl) – 3, 3 – dimethyldecane
Direct bromination of benzene with excess reagent results in the formation of ____________.
Dow's process is used for the synthesis of an aromatic compound (X). Identify X.
Which of the following compounds is NOT obtained in thermal decomposition of propane in absence of air?
