Advertisements
Advertisements
Question
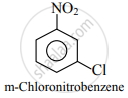
Predict the possible product of the following reaction.
Chlorination of nitrobenzene
Solution
The Nitro group is a meta directing group. So, chlorination of nitrobenzene gives m-chloronitrobenzene.
APPEARS IN
RELATED QUESTIONS
Predict the possible product of the following reaction.
sulphonation of chlorobenzene
Predict the possible product of the following reaction.
bromination of phenol
Identify the main product of the reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K]}\\
|\phantom{............}\\\ce{OH}\phantom{..........}
\end{array}\] _______.
Identify the main product of the reaction:
\[\ce{HC ≡ C - CH3 ->[H2][Pd-C/quinoline]}\] _______.
Name two reagents used for acylation of benzene.
Identify the compound (A) in the following reaction
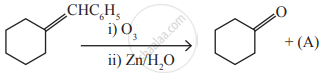
Which one of the following is non aromatic?
Some meta-directing substituents in aromatic substitution are given. Which one is most deactivating?
2 – butyne on chlorination gives ______.
Identify the compound A, B, C and D in the following series of reactions.

How is propyne prepared from an alkylene dihalide ?
Describe the mechanism of Nitration of benzene.
How does Huckel rule help to decide the aromatic character of a compound?
How will you convert ethyl chloride into ethane?

Identify A and B.
Complete the following:
\[\begin{array}{cc}
\ce{CH2 - CH2 ->[Zn/C2H5OH]}\\
|\phantom{.......}|\phantom{..............}\\
\ce{Br}\phantom{.....}\ce{Br}\phantom{..............}
\end{array}\]
When acetylene is passed through red hot iron tube at 873 K, ____________ is/are obtained as product/s.
−Cl group is ____________.
According to Huckel rule, a cyclic π molecular orbital formed by overlap of p orbitals must contain ____________ p electrons.
Which of the following is NOT alicyclic compound?
Direct bromination of benzene with excess reagent results in the formation of ____________.
The alkane formed on heating sodium butanoate with sodalime is ______.
Sodium benzoate on decarboxylation gives ____________.
α-butylene when subjected to hydroboration oxidation reaction, yields ____________.
Which of the following compounds on bromination yields ![]() ?
?
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH_3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..........................}\ce{CH3}\\\phantom{........................}|\\\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{......................................}\\
\ce{CH3}\phantom{....................................}\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
