Advertisements
Online Mock Tests
Chapters
2: Quantum Mechanical Model of Atom
3: Periodic Classification Of Elements
4: Hydrogen
5: Alkali and Alkaline Earth Metals
6: Gaseous State
7: Thermodynamics
8: Physical and Chemical Equilibrium
9: Solutions
10: Chemical bonding
11: Fundamentals of Organic Chemistry
12: Basic concept of organic reactions
▶ 13: Hydrocarbons
14: Haloalkanes and Haloarenes
15: Environmental Chemistry
![Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 11 TN Board chapter 13 - Hydrocarbons Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 11 TN Board chapter 13 - Hydrocarbons - Shaalaa.com](/images/chemistry-volume-1-and-2-english-class-11-tn-board_6:5f2b1b2038084cf381bfa42c826a928c.jpg)
Advertisements
Solutions for Chapter 13: Hydrocarbons
Below listed, you can find solutions for Chapter 13 of Tamil Nadu Board of Secondary Education Samacheer Kalvi for Chemistry - Volume 1 and 2 [English] Class 11 TN Board.
Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 11 TN Board 13 Hydrocarbons Evaluation [Pages 220 - 224]
Choose the best answer
The correct statement regarding the comparison of staggered and eclipsed conformations of ethane, is ______.
The eclipsed conformation of ethane is more stable than staggered conformation even though the eclipsed conformation has torsional strain.
The staggered conformation of ethane is more stable than eclipsed conformation, because staggered conformation has no torsional strain.
The staggered conformation of ethane is less stable than eclipsed conformation, because staggered conformation has torsional strain.
The staggered conformation of ethane is less stable than eclipsed conformation, because staggered conformation has no torsional strain.
\[\ce{C2H5Br + 2Na ->[dry ether]C4H10 + 2NaBr}\]
The above reaction is an example of which of the following.
Reimer Tiemann reaction
Wurtz reaction
Aldol condensation
Hoffmann reaction
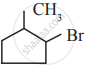
An alkyl bromide (A) reacts with sodium in ether to form 4, 5 – diethyloctane, the compound (A) is
CH3(CH2)3Br
CH3(CH2)5Br
CH3(CH2)3 CH(Br)CH3
\[\begin{array}{cc}\ce{CH3 - (CH2)2 - CH(Br) - CH2}\\\phantom{.........................}|\\
\phantom{............................}\ce{CH3}
\end{array}\]
The C – H bond and C – C bond in ethane are formed by which of the following types of overlap.
sp3 – s and sp3 – sp3
sp2 – s and sp2 – sp2
sp – sp and sp – sp
p – s and p – p
In the following reaction,

The major product obtained is
Which of the following is optically active?
2 – methyl pentane
citric acid
Glycerol
none of of these
The compounds formed at anode in the electrolysis of an aquous solution of potassium acetate are ______.
CH4 and H2
CH4 and CO2
C2H6 and CO2
C2H4 and Cl2
The general formula for cyclo alkanes.
CnHn
CnH2n
CnH2n-2
CnH2n+2
The compound that will react most readily with gaseous bromine has the formula (NEET) ______.
C3H6
C2H2
C4H10
C2H4
Which of the following compounds shall not produce propene by reaction with HBr followed by elemination (or) only direct elimination reaction (NEET).
∇
\[\ce{CH3 – CH2 – CH2 – OH}\]
\[\ce{H2C = C = O}\]
\[\ce{CH3 – CH2 – CH2Br}\]
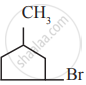
Which among the following alkenes on reductive ozonolysis produces only propanone?
2 – Methyl propene
2 – Methyl but – 2 – ene
2, 3 – Dimethyl but – 1- ene
2, 3 – Dimethyl but – 2 – ene
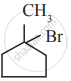
The major product formed when 2 – bromo – 2 – methyl butane is refluxed with ethanolic KOH is ______.
2 – methylbut – 2- ene
2 – methyl butan – 1 – ol
2 – methyl but – 1 – ene
2 – methyl butan – 2- ol
Major product of the below mentioned reaction is, \[\ce{(CH3 )2 C = CH2 ->[ICI]}\]
2 – chloro – 1 – iode – 2 – methyl propane
1 – chloro – 2 – iodo – 2 – methyl propane
1, 2 – dichloro – 2 – methyl propane
1, 2 – diiodo – 2 – methyl propane
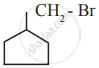
The IUPAC name of the following compound is
\[\begin{array}{cc}
\phantom{........}\ce{Cl}\phantom{......}\ce{CH2 - CH3}\\\backslash\phantom{......}/\\
\ce{C = C}\\/\phantom{......}\backslash\\
\ce{H3C}\phantom{......}\ce{I}
\end{array}\]
trans – 2- chloro – 3- iodo – 2- pentane
cis – 3- iodo – 4 chloro – 3 – pentane
trans – 3 – iodo – 4 – chloro – 3 – pentene
cis – 2 – chloro – 3 iodo – 2 – pentene
Cis – 2- butene and trans – 2 – butene are ______.
conformational isomers
structural isomers
configurational isomers
optical isomers
Identify the compound (A) in the following reaction
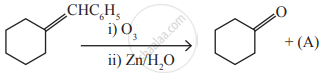
\[\begin{array}{cc}
\ce{CH2 - CH2 ->[(A)] CH ≡ CH}\\
|\phantom{.......}|\phantom{.................}\\
\ce{Br}\phantom{.....}\ce{Br}\phantom{.................}
\end{array}\], where A is,
Zn
conc. H2SO4
alc. KOH
dil. H2SO4
Consider the nitration of benzene using mixed con H2SO4 and HNO3 if a large quantity of KHSO4 is added to the mixture, the rate of nitration will be ______.
unchanged
doubled
faster
slower
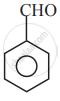
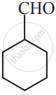
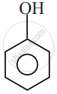
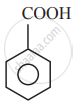
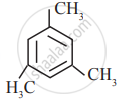
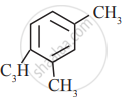
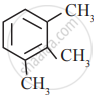
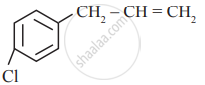
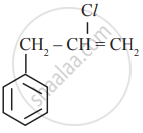
In which of the following molecules, all atoms are co-planar
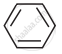
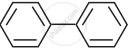
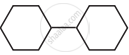
both
 and
and 
Propyne on passing through red hot iron tube gives
none of these
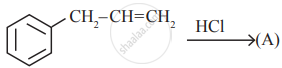 is
is
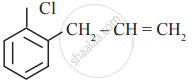
both
 and
and 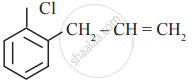
Which one of the following is non aromatic?
Which of the following compounds will not undergo Friedal – crafts reaction easily ? (NEET)
Nitro benzene
Toluene
Cumene
Xylene
Some meta-directing substituents in aromatic substitution are given. Which one is most deactivating?
– COOH
– NO2
-C ≡ N
-SO3H
Which of the following can be used as the halide component for friedal - crafts reaction?
Chloro benzene
Bromo benzene
Chloro ethene
Isopropyl chloride
An alkane is obtained by decarboxylation of sodium propionate. Same alkane can be prepared by ______.
Catalytic hydrogenation of propene
action of sodium metal on iodomethane
reduction of 1 – chloro propane
reduction of bromomethane
Which of the following is aliphatic saturated hydrocarbon?
C8H18
C9H18
C8H14
All of these
Identify the compound 'Z' in the following reaction
\[\ce{C2H6O ->[Al2O3][623 K] X ->[O3] Y ->[Zn/H2O] (Z)}\]
Formaldehyde
Acetaldehyde
Formic acid
none of these
Peroxide effect (Kharasch effect) can be studied in case of ______.
Oct – 4 – ene
hex – 3 – ene
pent – 1 – ene
but – 2 – ene
2 – butyne on chlorination gives ______.
1 – chloro butane
1, 2 – dichloro butane
1, 1, 2, 2 – tetrachlorobutane
2, 2, 3, 3 – tetra chloro butane
Write brief answer to the following questions.
Give IUPAC name for the following compound.
\[\ce{CH3 –CH=CH–CH=CH–C≡C–CH3}\]
Give IUPAC name for the following compound.
\[\begin{array}{cc}\ce{C2H5}\phantom{.}\ce{CH3}\phantom{.......}\\|\phantom{.....}|\phantom{.........}\\
\ce{CH3 - C - C - C ≡ C - CH3}\\
|\phantom{.....}|\phantom{.........}\\
\ce{CH3}\phantom{..}\ce{H}\phantom{.........}
\end{array}\]
Give IUPAC name for the following compound.
\[\ce{(CH3)3 C – C ≡ C – CH (CH3)2}\]
Give IUPAC name for the following compound.
Ethyl isopropyl acetylene
Give IUPAC name for the following compound.
\[\ce{CH ≡ C – C ≡ C – C ≡ CH}\]
Identify the compound A, B, C and D in the following series of reactions.

Write short notes on ortho, para directors in aromatic electrophilic substitution reactions.
How is propyne prepared from an alkylene dihalide ?
An alkyl halide with molecular formula C6H13Br on dehydro halogenation gave two isomeric alkenes X and Y with molecular formula C6H12. On reductive ozonolysis, X and Y gave four compounds CH3COCH3, CH3CHO, CH3CH, CHO and (CH3)2 CHCHO. Find the alkyl halide.
Describe the mechanism of Nitration of benzene.
How does Huckel rule help to decide the aromatic character of a compound?
Suggest the route for the preparation of the following from benzene.
3 – chloro nitrobenzene
Suggest the route for the preparation of the following from benzene.
4 – chlorotoluene
Suggest the route for the preparation of the following from benzene.
Bromo benzene
Suggest the route for the preparation of the following from benzene.
m - dinitro benzene
Suggest a simple chemical test to distinguish propane and propene.
What happens when Isobutylene is treated with acidified potassium permanganate?
How will you convert ethyl chloride into ethane?
How will you convert ethyl chloride into n – butane?
Describe the conformers of n – butane.
Write the chemical equations for combustion of propane.
Explain Markow nikoff’s rule with suitable example.
What happens when ethylene is passed through cold dilute alkaline potassium permanganate.
Write the structure of the following alkanes.
2, 3 – Dimethyl – 6 – (2 – methyl propyl) decane
Write the structure of the following alkanes.
5 – (2 – Ethyl butyl) – 3, 3 – dimethyldecane
Write the structure of the following alkanes.
5 – (1, 2 – Dimethyl propyl) – 2 – methylnonane
How will you prepare propane from a sodium salt of fatty acid?

Identify A and B.
Complete the following:
\[\ce{2 - butyne ->[Lindlar Catalyst]}\]
Complete the following:
\[\ce{CH2 = CH2 ->[I2]}\]
Complete the following:
\[\begin{array}{cc}
\ce{CH2 - CH2 ->[Zn/C2H5OH]}\\
|\phantom{.......}|\phantom{..............}\\
\ce{Br}\phantom{.....}\ce{Br}\phantom{..............}
\end{array}\]
Complete the following:
\[\ce{CaC2 ->[H2O]}\]
How will distinguish 1 – butyne and 2 – butyne?
Solutions for 13: Hydrocarbons
![Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 11 TN Board chapter 13 - Hydrocarbons Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 11 TN Board chapter 13 - Hydrocarbons - Shaalaa.com](/images/chemistry-volume-1-and-2-english-class-11-tn-board_6:5f2b1b2038084cf381bfa42c826a928c.jpg)
Samacheer Kalvi solutions for Chemistry - Volume 1 and 2 [English] Class 11 TN Board chapter 13 - Hydrocarbons
Shaalaa.com has the Tamil Nadu Board of Secondary Education Mathematics Chemistry - Volume 1 and 2 [English] Class 11 TN Board Tamil Nadu Board of Secondary Education solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. Samacheer Kalvi solutions for Mathematics Chemistry - Volume 1 and 2 [English] Class 11 TN Board Tamil Nadu Board of Secondary Education 13 (Hydrocarbons) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. Samacheer Kalvi textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in Chemistry - Volume 1 and 2 [English] Class 11 TN Board chapter 13 Hydrocarbons are Introduction and Classification of Alkanes, Alkanes, Alkenes, Alkynes, Aromatic Hydrocarbons.
Using Samacheer Kalvi Chemistry - Volume 1 and 2 [English] Class 11 TN Board solutions Hydrocarbons exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in Samacheer Kalvi Solutions are essential questions that can be asked in the final exam. Maximum Tamil Nadu Board of Secondary Education Chemistry - Volume 1 and 2 [English] Class 11 TN Board students prefer Samacheer Kalvi Textbook Solutions to score more in exams.
Get the free view of Chapter 13, Hydrocarbons Chemistry - Volume 1 and 2 [English] Class 11 TN Board additional questions for Mathematics Chemistry - Volume 1 and 2 [English] Class 11 TN Board Tamil Nadu Board of Secondary Education, and you can use Shaalaa.com to keep it handy for your exam preparation.