Advertisements
Advertisements
Question
Write short notes on ortho, para directors in aromatic electrophilic substitution reactions.
Solution
- All the activating groups are ‘ortho-para’ directors.
Example: – OH, – NH2, -NHR, -NHCOCH3, -OCH3 -CH3 – C2H5 etc. - Let us consider the directive influences of phenolic (-OH) group. Phenol is the resonance hybrid of following structures.
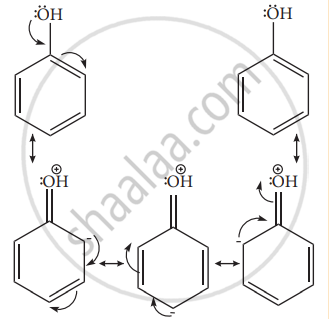
- In these resonance structures, the (-) charge residue is present on ortho and para position of ring structure. It is quite evident that the lone pair of electron on the atom which is attached to the ring involves in resonance and makes the ring more electron rich than benzene. The electron density at ortho and para positions increases as compared to the meta position. Therefore phenolic group activates the benzene ring for electrophilic attack at ‘ortho’ and ‘para positions and hence -OH group is an ortho-para director and activator.
- In aryl halides, the strong -I effect of the halogens (electron withdrawing tendency) decreases the electron density of benzene ring, thereby deactivating for electrophilic attack. However the presence of lone pair on halogens involved in the resonance with pi electrons of benzene ring, increases electron density at ortho and para position. Hence the halogen group is an ortho-para director and deactivator.
APPEARS IN
RELATED QUESTIONS
Identify giving reason whether the following compound is aromatic or not.

How is propyne prepared from an alkylene dihalide ?
Suggest the route for the preparation of the following from benzene.
3 – chloro nitrobenzene

Identify A and B.
Complete the following:
\[\begin{array}{cc}
\ce{CH2 - CH2 ->[Zn/C2H5OH]}\\
|\phantom{.......}|\phantom{..............}\\
\ce{Br}\phantom{.....}\ce{Br}\phantom{..............}
\end{array}\]
Which of the following is an aromatic compound?
α-butylene when subjected to hydroboration oxidation reaction, yields ____________.
Which of the following reagents can bring about following conversion?
\[\ce{But-1-ene -> Butan-2-ol}\]
The treatment of benzene with iso - butene in the presence of sulphuric acid gives:
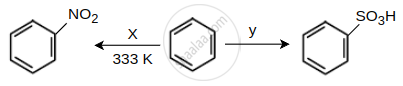
Identify the X and Y in the following reactions.