Advertisements
Advertisements
Question
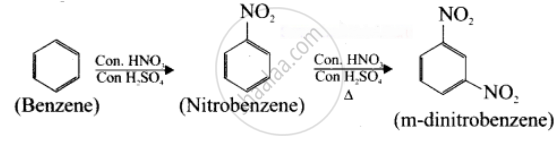
Suggest the route for the preparation of the following from benzene.
m - dinitro benzene
Solution
Benzene undergo twice the time nitration to give m-dinitrobenzene.
APPEARS IN
RELATED QUESTIONS
Identify the main product of the reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K]}\\
|\phantom{............}\\\ce{OH}\phantom{..........}
\end{array}\] _______.
Identify giving reason whether the following compound is aromatic or not.
Describe the mechanism of Nitration of benzene.
When acetylene is passed through red hot iron tube at 873 K, ____________ is/are obtained as product/s.
Which of the following is NOT alicyclic compound?
Direct bromination of benzene with excess reagent results in the formation of ____________.
α-butylene when subjected to hydroboration oxidation reaction, yields ____________.
Which of the following molecules has shortest C - C bond length?
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
