Advertisements
Advertisements
Question
How will you prepare propane from a sodium salt of fatty acid?
Solution
\[\ce{CH3 \underset{\text{(Sodium Salt of butyric acid)}}{- CH2 - CH2 - COONa} ->[NaOH/CaO] \underset{\text{(Propane)}}{CH3 - CH2} - CH3 + Na2CO3}\]
Sodium salt of butyric acid on heating with sodalime gives propane.
APPEARS IN
RELATED QUESTIONS
Name the following:
The hydrocarbon said to possess carcinogenic property.
Identify A, B, C in the following reaction sequence:
\[\ce{CH3 - CH = CH2 ->[Br2/CCl4][room temperature] A ->[Zn] B ->[dil. alkaline][KMnO4] C}\]
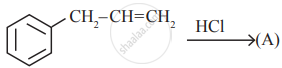 is
is
Write short notes on ortho, para directors in aromatic electrophilic substitution reactions.
How is propyne prepared from an alkylene dihalide ?
Write the chemical equations for combustion of propane.
How does Huckel rule help to decide the aromatic character of a compound?
The alkane formed on heating sodium butanoate with sodalime is ______.
α-butylene when subjected to hydroboration oxidation reaction, yields ____________.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{...............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
