Advertisements
Advertisements
Question
Complete the following reaction:
\[\ce{(C6H5CH2)2Cd + 2CH3COCI}\]
Solution
Treatment of acyl chlorides with dialkylcadmium gives a ketone.
\[\ce{2 CH3 - CO - CI + (C6H5- CH2)2 Cd -> \underset{\text{1-Phenylpropan-2-one}}{2 CH3-CO-CH2-C6H5} + CdCI2}\]
APPEARS IN
RELATED QUESTIONS
What is the action of following reagents on ethanoic acid?
SOCl2/heat
The formation of aldehyde from an alkyl cyanide using SnCl2/conc.HCl is known as ____________.
In the formation of an aldol, α-carbon atom of one aldehyde molecule attaches to ____________ of another aldehyde molecule.
Which among the following compounds is obtained when benzene is treated with CO and HCl in presence of catalyst anhydrous AlCl3 and Cu2Cl2 under high pressure?
Which of the following is obtained by hydrogenation of benzoyl chloride in presence of Pd on BaSO4?
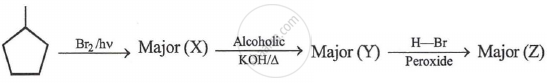
Z is:
What is the action of hydrogen cyanide on the following?
Benzaldehyde
Write Stephen reaction for the preparation of aldehyde.
How butanone is prepared from alcohol?
How are the following compound obtained by hydration of alkenes?
tert-butyl alcohol
