Advertisements
Advertisements
Question
Complete the following table for homologous series of Alkenes.
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Ethene | C2H4 | CH2 = CH2 | 2 | 0 | -102 |
| Propene | C3H6 | CH3–CH = CH2 | 3 | 1 | -48 |
| 1-Butene | C4H8 | CH3–CH2–CH = CH2 | ______ | ______ | -6.5 |
| 1-Pentene | C5H10 | CH3–CH2–CH2–CH = CH2 | ______ | ______ | 30 |
Advertisements
Solution
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Ethene | C2H4 | CH2 = CH2 | 2 | 0 | -102 |
| Propene | C3H6 | CH3–CH = CH2 | 3 | 1 | -48 |
| 1-Butene | C4H8 | CH3–CH2–CH = CH2 | 4 | 2 | -6.5 |
| 1-Pentene | C5H10 | CH3–CH2–CH2–CH = CH2 | 5 | 3 | 30 |
APPEARS IN
RELATED QUESTIONS
Classify the following carbon compounds into two homologous series and name them.
C3H4, C3H6, C4H6, C4H8, C5H8, C5H10
Write the name and formula of the 2nd member of homologous series having general formula CnH2n – 2.
Write the molecular formula of first two members of homologous series having functional group −Br.
Write the molecular formula of first two members of homologous series having functional group – OH
Fill in the following blank with suitable word:
Carbon compounds have usually ... melting points and boiling points because they are ...... in nature.
What is the difference between two consecutive homologues:
(1) in terms of molecular mass?
(2) in terms of number and kind of atoms per molecule?
Give the molecular formula of one homologue of the following:
C3H6
Give the molecular formula of one homologue of each of the following:
C2H6
Define a homologous series. Give the name and structural formula of one homologue of the following:
CH3OH
The molecular formula of an organic compound is C18H36. Name its homologous series.
The molecular formula of a homologue of butane is:
(a) C4H8
(b) C3H6
(c) C4H6
(d) C3H8
The molecular formula of the third member of the homologous series of ketones is:
(a) C4H8O
(b) C3H6O
(c) C5H10O
(d) C6H12O
A colourless organic liquid X of molecular formula C2H4O2 turns blue litmus to red. Another colourless organic liquid Y of molecular formula C3H6O has no action on any litmus but it is used as a nail polish remover. A yet another colourless organic liquid Z of molecular formula C2H6O has also no action on litmus but it is used in tincture of iodine.
(a) Name the liquid X. To which homologous series does it belong? Give the name of another member of this homologous series.
(b) Name the liquid Y. To which homologous series does it belong? Write the name of another member of this homologous series.
(c) Can you name an organic compound having the same molecular formula as liquid Y but which belongs to a different homologous series? What is this homologous series?
(d) Name the liquid Z. To which homologous series does it belong? Write the name of another member of this homologous series.
Succeeding members of a homologous series differ by ______.
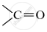 .
.
Define homologous series of organic compounds. List its two characteristics. Write the name and formula of the first member of the series of alkenes.
Distinguish between homologous organs and analogous organs. In which category would you place wings of a bird and wings of a bat? Justify your answer giving a suitable reason.
Complete the following table for homologous series of alcohols.
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Methanol | CH4O | CH3-OH | 1 | 1 | 63 |
| Ethanol | C2H6O | CH3–CH2-OH | 2 | 2 | 78 |
| Propanol | C3H8O | CH3–CH2–CH2-OH | ______ | ______ | 97 |
| Butanol | C4H10O | CH3–CH2–CH2–CH2–OH | ______ | ______ | 118 |
Which of the following pairs can be the successive members of a homologous series?
