Advertisements
Advertisements
Question
Describe the graphical representation of first order reaction.
Solution
Rate constant for first order reaction is,
kt = ln `((["A"_0])/(["A"]))`
kt = In [A0] – In [A]
In [A] = In [A0] – kt
y = c + mx
If we follow the reaction by measuring the concentration of the reactants at regular time intervals ‘t’, a plot of ln [A] against ‘t’ yields a straight line with a negative slope. From this, the rate constant is calculated.
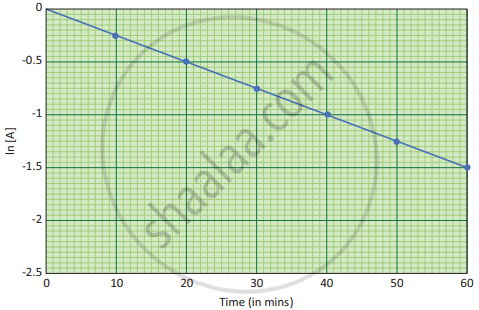
A plot of ln [A] Vs t for a first order reaction, \[\ce{A -> product}\] with initial concentration of [A] = 1.00 M and k = 2.5 × 10−2 min−1
APPEARS IN
RELATED QUESTIONS
Derive an expression for the relation between half-life and rate constant for first-order reaction.
For a first order reaction \[\ce{A ->Product}\] with initial concentration x mol L−1, has a half life period of 2.5 hours. For the same reaction with initial concentration `("x"/2)` mol L−1 the half life is
For a first-order reaction, the rate constant is 6.909 min−1 the time taken for 75% conversion in minutes is
What is the value of rate constant of first order reaction, if it takes 15 minutes for consumption of 20% of reactants?
The rate of the reaction \[\ce{A + B -> C}\] is 3.6 × 10−2 mol dm−3 s−1 when [A] = 0.3 mol dm−3 and [B] = 0.2 mol dm−3. Calculate k if reaction is first order in A and zero order in B.
A first order reaction is 25% completed in 40 minutes. What is the rate constant k tor the reaction?
The slope of a graph, log [A]t versus 't' for a first order reaction is −2.5 × 10−3 s−1. The rate constant for the reaction is ____________.
The initial concentration of reactant (A) is 2 mol dm−3 for a zero order reaction \[\ce{A -> B}\]. The rate constant (k) is related to its half-life `("t"_(1//2))` by the equation:
The integrated rate equation is Rt = log C0 – log Ct, then the straight-line graph is obtained by plotting.
The rate constant and half-life of a first order reaction are related to each other as ______.
