Advertisements
Advertisements
Question
Discuss briefly, giving an example of the role of coordination compounds in extraction/metallurgy of metals.
Solution
Some important extraction processes of metals, like those of silver and gold, make use of complex formation. Gold, for example, combines with cyanide in the presence of oxygen and water to form the coordination entity [Au(CN)2 ]− in aqueous solution. Gold can be separated in metallic form from this solution by the addition of zinc.
APPEARS IN
RELATED QUESTIONS
Discuss briefly, giving an example of the role of coordination compounds in medicinal chemistry.
Using IUPAC norms write the formulae for Potassium trioxalatoaluminate(III)
Using IUPAC norms write the formulae for Dichloridobis(ethane-1,2-diamine)cobalt(III)
Write the structures of the main products in the following reactions :
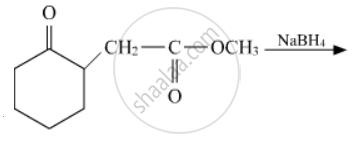
Discuss briefly, giving an example of the role of coordination compounds in biological systems.
Account for the following:
Ti(IV) is more stable than the Ti (II) or Ti(III).
The π - bonded organometallic compound which has ethene as one of its component is ______.
What is meant by the chelate effect? Give an example.
What is meant by the chelate effect? Give an example.
What is meant by the chelate effect? Give an example.
What is meant by the chelate effect? Give an example.
What is meant by the chelate effect? Give an example.
Discuss briefly, giving an example of the role of coordination compounds in analytical chemistry.
