Advertisements
Advertisements
Question
Discuss the mechanical equilibrium.
Solution
Mechanical equilibrium: Consider a gas container with the piston. When some mass is placed on the piston, it will move downward due to downward gravitational force, and after certain humps and jumps the piston will come to rest at a new position. When the downward gravitational force given by the piston is balanced by the upward force exerted by the gas, the system is said to be in mechanical equilibrium. A system is said to be in mechanical equilibrium if no unbalanced force acts on the thermodynamic system or on the surrounding by a thermodynamic system.
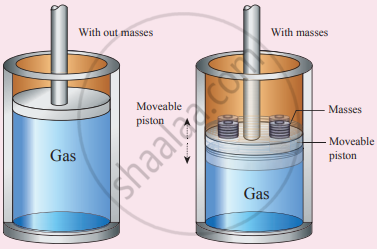
Mechanical equilibrium
APPEARS IN
RELATED QUESTIONS
A gas contained in a cylinder fitted with a frictionless piston expands against a constant external pressure of 1 atm from a volume of 5 liters to a volume of 10 liters. In doing so it absorbs 400J of thermal energy from its surroundings. Determine the change in the internal energy of the system.
A group of objects that can form a unit which may have the ability to exchange energy with its surrounding is called what?
Classify and explain the thermodynamic system.
What are the different types of thermodynamic systems?
What is meant by ‘thermal equilibrium’?
What is mean by state variable? Give example.
Discuss the chemical equilibrium.
Discuss the thermodynamic equilibrium.
An insulated container of gas has two chambers separated by an insulating partition. One of the chambers has volume V1 and contains ideal gas at pressure P1 and temperature T1. The other chamber has volume V2 and contains ideal gas at pressure P2 and temperature T2. If the partition is removed without doing any work on the gases, calculate the final equilibrium temperature of the container.
What are surroundings in thermodynamics?
