Advertisements
Advertisements
Question
Distinguish between the given pairs of compounds using the test given within brackets:
Sodium nitrate and sodium sulphite (using dilute sulphuric acid).
Solution
Sodium nitrate on treatment with dilute sulphuric acid gives sodium bisulphate and nitric acid.
\[\ce{NaNO3 + H2SO4 -> NaHSO4 + HNO3}\]
Sodium sulphite treated with dilute sulphuric acid gives sodium sulphate and sulphur dioxide.
\[\ce{Na2SO3 + H2SO4 → Na2SO4 + H2O + SO2}\]
APPEARS IN
RELATED QUESTIONS
Write a balanced chemical equation for the following:
Action of concentrated sulphuric acid on Sulphur.
Write balanced chemical equations to show how SO3 is converted to Sulphuric acid in the contact process.
Identify the acid in each case:
The acid which produces sugar charcoal from sugar
Distinguish between the given pairs of compounds using the test given within brackets:
Dilute sulphuric acid and dilute hydrochloric acid (using barium chloride solution)
Why is Concentrated sulphuric acid kept in air tight bottles?
Give reason for the following:
When concentrated sulphuric acid is added to blue crystalline copper sulphate, it turns powdery white.
How are the following conversion brought about? Give equation and condition:
Oxalic acid to carbon monoxide.
Write the equation for the laboratory preparation of the following salts, using sulphuric acid.
Iron (II) sulphate from iron.
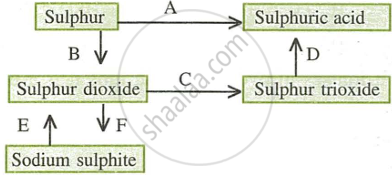
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
Write a balanced equation for the following conversion:
Lead sulphate from lead nitrate and sulphuric acid.
