Advertisements
Advertisements
Question
Distinguish between the three states of matter—solid, liquid, and gas on the basis of their molecular models
Solution
Solids:
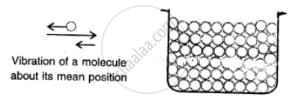
Here the molecules are very tighty packed that there is no or very less inteimolecular space and there is high intermolecular force of attraction (force of cohesion).
The molecules do not move about their mean position and thus solids have a definite shape and volume.
Liquids:
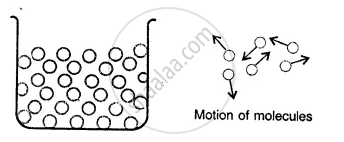
Here the molecules are less tightly packed as compared to solids and also there is lesser force of intermolecular attraction. The intermolecular distance is greater than that in the solids. Thus, they donot have a definite shape but acquire the shape of the vessel in which they are contained but have a definite volume at a given temperature.
Gases:
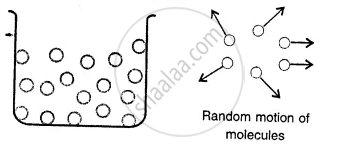
Here the molecules are far apart from each other i.e. have the greatest intermolecular distance which result into the weakest intermolecular forces of attraction. The molecules as are not bound by any strong force move about freely and thus gases do not have a definite shape and also do not have any definite volume.
APPEARS IN
RELATED QUESTIONS
Give the molecular model for a solid and use it to explain why a solid has a definite volume and a definite shape.
Describe the molecular model for a liquid. I-low does it explain that a liquid has no definite shape, but has a definite Volume?
Differentiate between the following:
Liquids and gases
Fill in the blank
When a gas is cooled, its molecules .............. energy.
Name the phenomenon which cause the following change.
Disappearance of camphor.
Multiple choice question.
Which of the following is a property of the liquids?
Name the three states of matter and define them.
State what would you observe if
(a) sugar is added to pebbles take in a plastic beaker
(b) sand is added to glass balls in a beaker. What would you conclude from this imaginative demonstration?
Complete the statement given below by selecting the correct word.
The conversation of vapour into a liquid is called ____.
For removing grains from stalks, the ______ method is used.
