Advertisements
Advertisements
Question
Draw a graph showing the variation of the number of particles scattered (N) with the scattering angle θ in the Geiger-Marsden experiment. Why only a small fraction of the particles are scattered at θ > 90°?
Solution
Graph showing the variation of the number of particles scattered (N) with the scattering angle (θ):
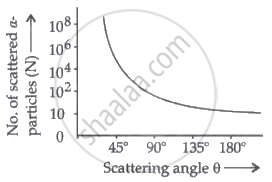
The strongly repulsive force exerted by the positively charged nucleus causes the scattering of α-particles at θ > 90°. Because the nucleus is so small, there are fewer distributed α-particles.
RELATED QUESTIONS
Thorium 90Th232 is disintegrated into lead 82Pb200. Find the number of α and β particles emitted in disintegration.
A classical atom based on ____________ is doomed to collapse.
Suppose you are given a chance to repeat the alpha-particle scattering experiment using a thin sheet of solid hydrogen in place of the gold foil. (Hydrogen is a solid at temperatures below 14 K.) What results do you expect?
Answer the following question, which help you understand the difference between Thomson’s model and Rutherford’s model better.
Keeping other factors fixed, it is found experimentally that for small thickness t, the number of α-particles scattered at moderate angles is proportional to t. What clue does this linear dependence on t provide?
Alpha particles used in Geiger-Marsden experiment were obtained from ______.
The equation of trajectory of a projectile is given by y = `"x"/sqrt3 - "gx"^2/20`, where x and y are in metres. The maximum range of the projectile is:
Nucleolus of an atom of mass No. 24 and charge no. 11 consists of
Assume that there is no repulsive force between the electrons in an atom but the force between positive and negative charges is given by Coulomb’s law as usual. Under such circumstances, calculate the ground state energy of a He-atom.
The energy levels of a certain atom for first, second and third levels are E, 4E/3 and 2E, respectively. A photon of wavelength λ is emitted for a transition 3 `→` 1. What will be the wavelength of emission for transition 2 `→` 1?
How is the size of a nucleus found experimentally? Write the relation between the radius and mass number of a nucleus.
