Advertisements
Advertisements
Question
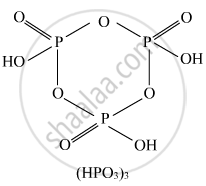
Draw the structures of the following molecules: (HPO3)3
Solution
(HPO3)3, cyclic structure
APPEARS IN
RELATED QUESTIONS
Draw the structure of: Orthophosphoric acid
Draw the structure of: Pyrophosphoric acid
Write balanced chemical equations for Action of ozone on hydrogen peroxide.
\[\begin{array}{cc}
\phantom{.}\ce{OH}\\
|\phantom{..}\\
\ce{O=P-OH}\\
|\phantom{..}\\
\ce{OH}
\end{array}\]
is the structure of:-
Nitrogen forms N2, but phosphorus is converted into P4 from P, the reason is:
Pentavalence in phosphorus is more stable when compared to that of nitrogen even though they belong to the same group. This is due to ____________.
Which of the following elements does not form stable diatomic molecules?
Which gas cannot be collected over water?
On heating with concentrated \[\ce{NaOH}\] solution in an inert atmosphere of \[\ce{CO2}\], white phosphorus gives a gas. Which of the following statement is incorrect about the gas?
What is the basicity of \[\ce{H3PO4}\]?
