Advertisements
Advertisements
Question
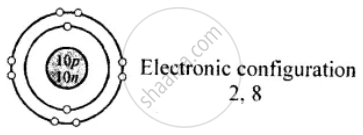
Draw the diagrams representing the atomic structures of the following: Neon
Solution
Neon \[\ce{^{20}_{10}Ne}\]
APPEARS IN
RELATED QUESTIONS
If bromine atom is available in the form of, say, two isotopes `""_35^79Br (49.7%)" and """_35^81Br (50.3%)`, calculate the average atomic mass of bromine atom.
Write the names of `"Ca"("NO"_3)_2` compounds :
Name the unit in which the radius of an atom is usually expressed.
Give brief information of three subatomic particles with reference to electrical charge, mass and location.
Define the following term:
Atom
Electron, proton, neutron are the types of ______ in an atom.
Elements X and Y have 1 and 7 electrons in their outermost shell respectively.
Which element will lose electron?
The smallest particle of matter that can exist by itself is ______.
Define the following term with an example.
Metalloid
The graphite refill used is pencil is made of element called ______.
