Advertisements
Advertisements
Question
During the preparation of hydrogen chloride gas on a humid day, the gas is usually passed through the guard tube containing calcium chloride. The role of calcium chloride taken in the guard tube is to ______
Options
absorb the evolved gas
moisten the gas
absorb moisture from the gas
absorb Cl– ions from the evolved gas
Solution
During the preparation of hydrogen chloride gas on a humid day, the gas is usually passed through the guard tube containing calcium chloride. The role of calcium chloride taken in the guard tube is to absorb moisture from the gas.
Explanation -
CaCl2 absorbs moisture to keep the gas in dry state. Otherwise, it will dissolve in moisture to form hydrochloric acid.
APPEARS IN
RELATED QUESTIONS
When acids react with metal, which gas is liberated?
Dry HCl gas does not change the colour of dry blue litmus paper. Why?
Equal lengths of magnesium ribbons are taken in test tubes A and B. Hydrochloric acid (HCl) is added to test tube A, while acetic acid (CH3COOH) is added to test tube B. In which test tube will the fizzing occur more vigorously, and why?
What ions are present in the solutions of following substances? (write the symbols only)
Sodium hydroxide
Name the product formed when Cl2 and H2 produced during the electrolysis of brine are made to combine.
The table given below shows the reaction of a few elements with acids and bases to evolve Hydrogen gas.
| Element | Acid | Base |
| A |
✗
|
✗ |
| B |
✓
|
✓ |
| C | ✓ | ✗ |
| D | ✓ | ✓ |
Which of these elements form amphoteric oxides?
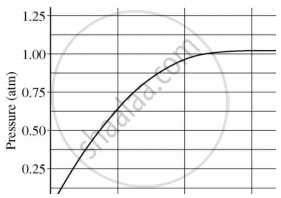
A student added 10 g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
Calcium phosphate is present in tooth enamel. Its nature is
Are all acids corrosive in nature? Name a few acids which are non-corrosive and may be part of our food.
For each of the salt: A, B, C and D, suggest a suitable method of its preparation.
C is a soluble salt of copper.
