Advertisements
Advertisements
Question
Explain inductive effect with suitable example.
Long Answer
Solution
- Inductive effect is defined as the change in the polarisation of a covalent bond due to the presence of adjacent bonds, atoms or groups in the molecule. This is a permanent phenomenon.
- Let us explain the inductive effect by considering ethane and ethyl chloride as examples. The \[\ce{C-C}\] bond in ethane is non-polar, while the \[\ce{C-C}\] bond in ethyl chloride is polar. We know that chlorine is more electronegative than carbon, and hence, it attracts the shared pair of electrons between \[\ce{C-Cl}\] in ethyl chloride towards itself. This develops a slight negative charge on chlorine and a slight positive charge on carbon to which chlorine is attached.
- To compensate for it, the \[\ce{C1}\] draws the shared pair of electrons between itself and \[\ce{C2}\]. This polarisation effect is called the inductive effect. This effect is greatest for the adjacent bonds, but they also are felt farther away. However, the magnitude of the charge separation decreases rapidly as we move away from \[\ce{C2}\] and is observed to be maximum for 2 carbons and almost insignificant after 4 bonds from the active group.
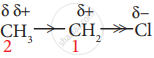
- It is important to note that the inductive effect does not transfer electrons from one atom to another, but the displacement effect is permanent. The inductive effect represents the ability of a particular atom or a group to either withdraw or donate electron density to the attached carbon. Based on this ability, the substituents are classified as +I groups and −I groups. Their ability to release or withdraw the electron through sigma covalent bond is called the +I effect and −I effect, respectively.
- Highly electronegative atoms and groups with an atom carrying a positive charge are electron-withdrawing groups, or −I groups. Example: \[\ce{-F , -Cl, -COOH, -NO2 NH2}\]
- The higher the electronegativity of the substituent, the greater the -I effect. The order of the -I effect of some groups are given below. \[\ce{NH3 > NO2 > CN > SO3H > CHO > CO > COOH > COCl > CONH2 > F > Cl > Br > I > OH > OR > NH2 > C6H5 > H}\]
- Highly electropositive atoms and atoms are groups that carry a negative charge and are electron donating or +I groups.
Example:
Alkali metals, alkyl groups such as methyl, ethyl, negatively charged groups such as \[\ce{CH3O–, C2H5O–, COO–}\] etc. - The lesser the electronegativity of the elements, the greater is the +1 effect. The relative order of +1 effect of some alkyl groups is given below.
\[\ce{-C(CH3)3 > -CH(CH3)2 > -CH2CH3 > -CH3}\]
shaalaa.com
Basic Concept of Organic Reactions
Is there an error in this question or solution?
Chapter 12: Basic concept of organic reactions - Evaluation [Page 176]
APPEARS IN
RELATED QUESTIONS
Decreasing order of nucleophilicity is ______.
Which of the following species is not electrophilic in nature?
Homolytic fission of covalent bond leads to the formation of ______.
Which of the following species does not exert a resonance effect?
- I effect is shown by ______.
Which of the following carbocation will be most stable?
Which of the following species does not acts as a nucleophile?
Write a short note on hyperconjucation.
Show the heterolysis of a covalent bond by using curved arrow notation and complete the following equation. Identify the nucelophile.
\[\ce{CH3 – O – CH3 + HI →}\]
Give example for the following type of organic reaction.
β - elimination
