Advertisements
Advertisements
Question
Explain solid solutions of metals and vacancy through aliovalent cations.
Solution
- Solid solutions of metals (alloys):
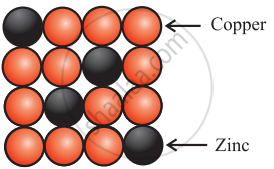
Brass
Brass is an alloy of Cu and Zn. In brass, host Cu atoms are replaced by impurity of Zn atoms. The Zn atoms occupy regular sites of Cu atoms as shown in the figure. - Formation of vacancy through aliovalent impurity:
Vacancies are created by the addition of impurities of aliovalent ions (that is, ions with oxidation state different from that of host ions) to an ionic solid.
e.g. Consider a small amount of SrCl2 impurity added to NaCl during its crystallization. The added Sr2+ ions (O.S. = +2) occupy some of the regular sites of Na+ host ions (O.S. = +1). In order to maintain electrical neutrality, every Sr2+ ion removes two Na+ ions. One of the vacant lattice sites created by removal of two Na+ ions is occupied by one Sr2+ ion. The other site of Na+ ion remains vacant as shown in the figure.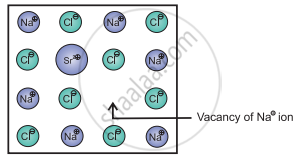
Vacancy through aliovalent ion
APPEARS IN
RELATED QUESTIONS
What are the consequences of Schottky defect?
What is an impurity defect? What are its types? Explain the formation of vacancies through aliovalent impurity with example.
Explain with diagram, the vacancy defect.
Give the classification of stoichiometric point defects.
Which of the following is an example of substitutional impurity defect?
When a cation or anion from ionic solid leaves its regular lattice site and moves to occupy an interstitial site, it is called as ____________.
Which among the following solids shows Frenkel defect?
Which of the following match is CORRECT?
(I) Frenkel defect: Electrical neutrality of the compound is preserved.
(II) Schottky defect: Density of the substance decreases.
(III) Schottky defect: Combination of vacancy defect and interstitial defect.
(IV) Frenkel defect: Small difference between size of cation and anion.
Schottky defect in crystals is observed when ____________.
Schottky defect is formed in ionic compounds with ____________.
AgBr shows which type of defect?
In which among the following solids, Schottky defect is not observed?
Explain the following term:
Substitutional impurity defect
Explain the following term:
Interstitial impurity defect
Explain Self interstitial defect in elemental solid.
Explain the interstitial defect.
What is a vacancy defect?
Give the disadvantages of a vacancy defect.
What are point defects?
