Advertisements
Advertisements
Question
Which electron displacement effect explains the following correct orders of acidity of the carboxylic acids?
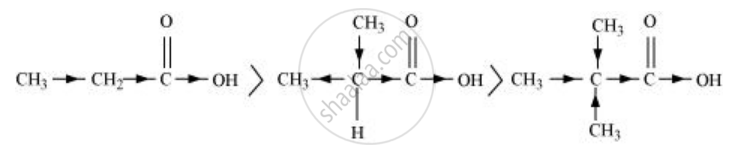
\[\ce{CH3CH2COOH > (CH3)2CHCOOH > (CH3)3C.COOH}\]
Short Answer
Solution
\[\ce{CH3CH2COOH > (CH3)2CHCOOH > (CH3)3C.COOH}\]
The order of acidity can be explained on the basis of the inductive effect (+I effect). As the number of alkyl groups increases, the +I effect also increases. With the increase in the +I effect, the basic strength also increases accordingly.
shaalaa.com
Fundamental Concepts in Organic Reaction Mechanism - Electron Displacement Effects in Covalent Bonds
Is there an error in this question or solution?
Chapter 12: Organic Chemistry - Some Basic Principles and Techniques - EXERCISES [Page 371]
