Advertisements
Advertisements
Question
Explain the reason:
Why 'food containing iron salts' should not be cooked in aluminium utensils?
Solution
Aluminium comes before iron in the metal activity series, so it can displace iron from iron salts; thus, food containing iron salts should not be cooked in aluminium utensils.
APPEARS IN
RELATED QUESTIONS
Answer the following questions with respect to the electrolytic process in the extraction of aluminum:
Identify the components of the electrolyte other than pure alumina and the role played by each
Name the solution used to react with Bauxite as a first step in obtaining pure aluminum oxide, in the Baeyer’s process
For sodium hydroxide, explain its significance in the extraction of aluminium.
The reason for using Aluminium in the alloy duralumin is ______.
What is the role of cryolite (NaAlF6) in the electrolytic reduction of alumina in the Hall's process?
Write balanced equation for the following reaction:
Aluminium powder is warmed with hot and concentrated caustic soda solution.
The sketch below illustrates the refin ing of aluminium by Hoope's process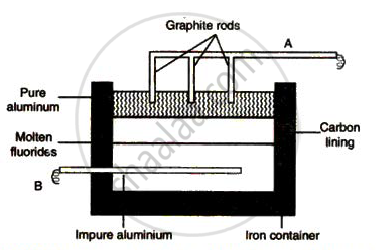
(a) Which of A and B is th e cathode and which one is the anode?
(b) What is the electroly te in the tank?
( c) What material is used for th e cathode?
Write the equation for the reaction where the aluminium oxide for the electrolytic extraction of aluminium is obtained by heating aluminium hydroxide.
Name the alloy used for the following purpose.
Surgical instruments
Name the alloy used for the following purpose.
Aircraft
