Advertisements
Advertisements
Question
The sketch below illustrates the refin ing of aluminium by Hoope's process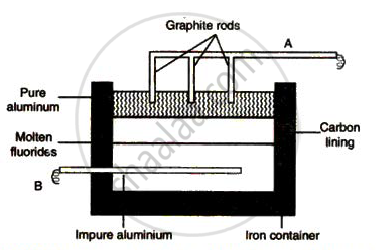
(a) Which of A and B is th e cathode and which one is the anode?
(b) What is the electroly te in the tank?
( c) What material is used for th e cathode?
Solution
(a) A is cathode and B is anode.
(b) Molten fluorides of Al, Na and Ba.
(c) Graphite rods.
APPEARS IN
RELATED QUESTIONS
For the substance given below, describe the role played in the extraction of aluminium.
Graphite
Explain why In the electrolysis of alumina using the Hall Heroult's Process the electrolyte is covered with powdered coke
Aluminium is said to be more reactive than iron, towards oxygen (or air) yet iron undergoes corrosion to a greater extent than aluminum. Explain.
Name the constituents of Solder.
Give reason for the following:
Nitric acid can be stored in aluminium containers.
For the substance listed below, explain its role in the extraction of aluminium: Bauxite
Aluminium is extracted from its chief ore bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction.
Write three balanced equations for the purification of bauxite by Hall's process.
The following question relate to the extraction of aluminium by electrolysis.
Give the equation for the reaction that takes place at the cathode
Name the following :
The compound formed by the reaction between calcium oxide and silica.
Fused alumina is reduced to aluminium by electrolytic reduction, since ______.
