Advertisements
Advertisements
Question
Give an example for each of the following statement
A compound in which two Covalent bonds are formed.
Solution
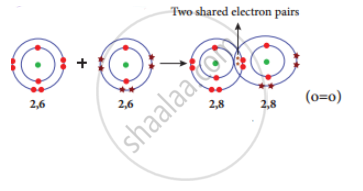
Formation of oxygen molecule (O2): Each oxygen atom has six valence electrons (2, 6). These two atoms achieve a stable electronic configuration (octet) by sharing two pair of electrons. Hence a double bond is formed in between the two atoms.
Formation of covalent bond in oxygen molecule.
APPEARS IN
RELATED QUESTIONS
Write any three features and give two examples of covalent compounds
Which inert gas does the H atom in HCl resemble in electron arrangement?
Why is graphite a good conductor of electricity but diamond is a non-conductor of electricity?
Explain the following briefly:
Sodium chloride dissolves in water but carbon tetra chloride is insoluble in water.
Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the options A, B, C and D given below:
X is likely to have a ______.
Draw the electron dot diagram and structure of :
methane
Two carbon atoms can always form one or two covalent bonds.
Molecular reactions are ______ in the covalent compound.
Explain dipole (polar) molecule by taking hydrogen chloride as an example.
