Advertisements
Advertisements
प्रश्न
Give an example for each of the following statement
A compound in which two Covalent bonds are formed.
उत्तर
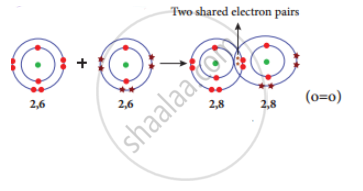
Formation of oxygen molecule (O2): Each oxygen atom has six valence electrons (2, 6). These two atoms achieve a stable electronic configuration (octet) by sharing two pair of electrons. Hence a double bond is formed in between the two atoms.
Formation of covalent bond in oxygen molecule.
APPEARS IN
संबंधित प्रश्न
What type of chemical bond is formed between carbon and bromine?
Compare the properties of ionic compounds and covalent compounds.
Give two example in following case:
Gaseous polar compounds
The covalent bond in which the electrons are shared equally between the combining atoms.
Taking hydrogen chloride and methane as examples, distinguish between a polar covalent bond and a non polar covalent bond.
Explain the following:
Covalent compounds have low melting and boiling point.
Explain the following:
Polar covalent compounds are good conductors of electricity.
Name a neutral covalent molecule which contains one lone pair of electrons.
Define a covalent bond.
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
 |
