Advertisements
Advertisements
Question
Give appropriate scientific reasons for The electrical conductivity of acetic acid is less in comparison to the electrical conductivity of dilute sulphuric acid at a given concentration.
Solution
Sulphuric acid is a strong acid compared to acetic acid. A strong acid has more ions than a weak one, and so, its solution will be a better electrical conductor than a weak acid. So, the electrical conductivity of acetic acid is less in comparison of electric conductivity of sulphuric acid.
APPEARS IN
RELATED QUESTIONS
Give balanced chemical equations for the action of sulphuric acid on the following:
Potassium hydrogen carbonate.
Name the products formed when hot and concentrated sulphuric acid reacts with Sulphur.
What would you observe in the following case?
Hot concentrated sulphuric acid is added to sodium chloride crystals.
How are the following conversion brought about? Give equation and condition:
Dilute sulphuric acid to hydrogen.
Complete the following sentence, choosing the correct word from the given options.
"Concentrated sulphuric acid is used in the laboratory preparation of nitric acid and hydrochloric acid because it is ____________ in comparison to these two acids."
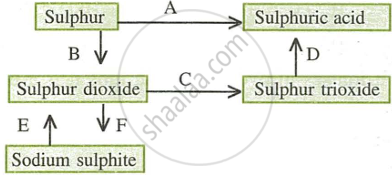
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
Write the equation of the following reaction:
Sulphur is heated with concentrated sulphuric acid
State your observation for the following case :
Dry red rose petals are placed in the jar of sulphur dioxide.
State one appropriate observation of the following:
Concentrated sulphuric acid is added dropwise to a crystal of hydrated copper sulphate.
Name the acid formed when sulphur dioxide dissolves in water.
