Advertisements
Advertisements
Question
State one appropriate observation of the following:
Concentrated sulphuric acid is added dropwise to a crystal of hydrated copper sulphate.
Solution
When conc. H2SO4 is added to a crystal of hydrated copper sulphate, it removes water of crystallisation from salt.
APPEARS IN
RELATED QUESTIONS
State one relevant observation for given reactions:
Addition of ethyl alcohol to acetic acid in the presence of concentrated Sulphuric acid
Identify the acid in the case:
The acid on mixing with lead nitrate solution produces a white precipitate, which is insoluble even on heating.
Name the following:
Products obtained by treating zinc with dilute sulphuric acid.
Choose the current answer from the options given below :
When conc. H2SO4 comes in contact with sugar, it becomes black due to
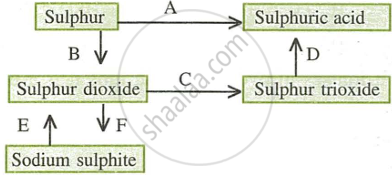
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
Give one equation to show the following properties of sulphuric acid : Dehydrating property
Concentrated sulphuric acid is both an oxidizing agent and a non-volatile acid. Write one equation each to illustrate the properties of sulphuric acid mentioned above.
Write the balanced chemical equation for the following conversion:
Lead sulphate from lead nitrate solution and dilute sulphuric acid.
Dilute sulphuric acid will produce a white precipitate when added to a solution of ______.
Identify the gas evolved and give the chemical test in the following case:
Dilute hydrochloric acid reacts with sodium sulphite.
