Advertisements
Advertisements
Question
Give one example of a chemical reaction.
Solution
Burning of magnesium ribbon in air to give white powder of magnesium oxide is an example of chemical reaction.
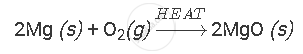
APPEARS IN
RELATED QUESTIONS
Methyl orange is used as an indicator. It shows colour change in acid, base and neutral substance. Tabulate your result as follows:
| Indicator | Colour change | Inference |
| Methyl Orange | No change | ...................... |
| .................... | Acid | |
| Yellow | ....................... |
2 mL of sodium hydroxide solution is added to a few pieces of granulated zinc metal taken in a test tube. When the contents are warmed, a gas evolves which is bubbled through a soap solution before testing. Write the equation of the chemical reaction involved and the test to detect the gas. Name the gas which will be evolved when the same metal reacts with dilute solution of a strong acid.
Write a balanced chemical equation for the process of photosynthesis giving the physical states of all the substances involved and the conditions of the reaction.
State one characteristic of the chemical reaction which takes place when dilute hydrochloric acid is added to sodium carbonate.
Write word equation for the following skeletal equation:
\[\ce{KClO3 -> KCl + O2}\]
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate the mass of salt formed.
Define: Endothermic reaction
Answer the following question.
What is observed when 2 mL of dilute hydrochloric acid is added to 1 g of sodium carbonate taken in a clean and dry test tube? Write a chemical equation for the reaction involved.
Which among the following statement(s) is(are) true? ‘Exposure of silver chloride to sunlight for a long duration turns grey due to
- the formation of silver by decomposition of silver chloride
- sublimation of silver chloride
- decomposition of chlorine gas from silver chloride
- oxidation of silver chloride
Dil. HCl is added to Zn granules. How will you prove that chemical change has taken place here? Support your response with two arguments.
