Advertisements
Advertisements
Question
Give reasons for the following:
N-N bond is weaker than P-P bond.
Solution
The nitrogen atom is smaller in size than the Phosphorus atom. The bond length of the N-N bond is smaller than that of the P-P bond. Due to this, the four non-bonding electrons of the two nitrogen atoms repel each other making the bond weaker.
This does not happen in the case of phosphorus because P-P bond length is more. So, the repulsion of non-bonding electrons is lesser than in N-N. So, the P-P sigma bond is stronger.
APPEARS IN
RELATED QUESTIONS
Give reasons for the following:
(i) Bond enthalpy of F2 is lower than that of Cl2.
(ii) PH3 has lower boiling point than NH3.
Identify 'A' and 'B' and rewrite the reactions
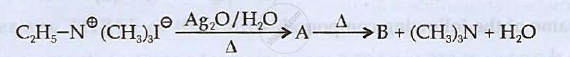
The ligand diethylene triamine is -
(a) monodentate
(b) bidentate
(c) tridentate
(d) tetradentate
Account for the following :
Bond angle in NH+4NH4+ is higher than NH3.
CH3 – mg – be is an organometallic compound due to
