Advertisements
Advertisements
Question
How is aluminium obtained from pure ore?
Solution
Aluminium is obtained from pure ore by Hall - Heroult's process as follows:
i. Electrolyte used :
a. Pure alumina
b. Cryolite
c. Aluminium fluoride
d. Calcium fluoride or Fluorspar
ii . Electrolytic cell : It is a rectangular steel tank with a thick inner lining of carbon .
iii . Electrodes used :
a. Cathode: inner lining of carbon
b. Anode: Thick carbon rods dipping into the fused electrolytes.
iv. Temperature of the electrolytic bath : 950°C
Reactions: On passage of electricity through electrolyte aluminium and oxide ions are formed.
At cathode: Aluminium ions get reduced as:
Al3+ + 3e- → Al
At anode: oxygen gas is liberated as:
O2- - 2e- → [ O ]
[ O ] + [ O ] → O2
The oxygen formed at anode oxidizes the carbon of the anode to carbon dioxide.
C + O2 → CO2
As a result the anode gets oxidized and it has to be replaced periodically.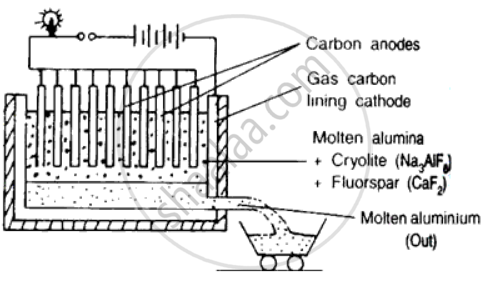
Figure: Extraction of aluminium from pure ore.
APPEARS IN
RELATED QUESTIONS
Write the equation for the reaction that occurs at the cathode during the extraction of aluminum by electrolysis.
For the substance given below, describe the role played in the extraction of aluminium.
Cryolite
Aluminium is a more active metal than iron, but suffers less corrosion. Why?
For bauxite, explain its significance in the extraction of aluminium.
| Aluminium is extracted from its chief ore, bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction. |
Name a chemical used for dissolving aluminium oxide. In which state is the chemical used?
Name the ore from which aluminium is extracted.
For the substance listed below, explain its role in the extraction of aluminium: Graphite
In order to obtain one tone of aluminium, the following inputs are required:
4 tones of bauxite, 150 Kg of sodium hydroxide and 600 Kg of graphite.
The aluminium compound in bauxite is aluminium oxide and the main impurity is iron (II) oxide. Aluminium is obtained by the electrolysis of aluminium oxide dissolved in cryolite.
(i) Name the process used for purification of bauxite.
(ii) Write the equation to show the action of heat on aluminium hydroxide.
Name the compound added to pure alumina to lower the fusion temperature during the electrolytic reduction of alumina.
Name the following :
The compound formed by the reaction between calcium oxide and silica.
