Advertisements
Advertisements
Question
For the substance given below, describe the role played in the extraction of aluminium.
Cryolite
Solution
- It lowers the fusion temperature from 2050°C to 950°C and enhances conductivity.
- This, in turn, improves the electrolyte's conductivity and reduces the need for electricity, which is the primary energy source for alumina reduction.
APPEARS IN
RELATED QUESTIONS
For the substance given below, describe the role played in the extraction of aluminium.
Graphite
Name the constituents of Duralumin.
Aluminium is extracted from its chief ore, bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction.
Write an equation for the reaction which takes place at the anode during the extraction of aluminium by the electrolytic process.
What is the role of cryolite (NaAlF6) in the electrolytic reduction of alumina in the Hall's process?
A to F below relate to the source and extraction of either zinc or aluminium.
- Bauxite
- Coke
- Cryolite
- Froth floatation
- Sodium hydroxide solution
- Zinc blende
Fill in the blanks using the most appropriate words from A to F:
(i) The ore from which aluminium is extracted must be treated with ______ so that pure aluminium oxide can be obtained.
(ii) Pure aluminium oxide is dissolved in ______ to make a conducting solution.
The sketch below illustrates the refin ing of aluminium by Hoope's process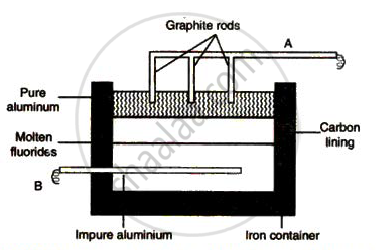
(a) Which of A and B is th e cathode and which one is the anode?
(b) What is the electroly te in the tank?
( c) What material is used for th e cathode?
Explain why :
Iron sheets are coated with zinc during galvanization.
Name the solution used to react with bauxite as a first step in obtaining pure aluminium oxide, in the Baeyer's process.
Fused alumina is reduced to aluminium by electrolytic reduction, since ______.
State true or false.
Bauxite is the main ore of aluminium.
