Advertisements
Advertisements
Question
Explain why :
Iron sheets are coated with zinc during galvanization.
Solution
Iron sheets are coated with zinc during galvanisation to prevent them from rusting.
APPEARS IN
RELATED QUESTIONS
For sodium hydroxide, explain its significance in the extraction of aluminium.
In order to obtain 1 tonne of aluminium, the following inputs are required: 4 tonnes of bauxite, 150 kg of sodium hydroxide and 600 kg of graphite. The aluminium compound in bauxite is aluminium oxide and the main impurity is iron (III) oxide. Aluminium is obtained by the electrolysis of aluminium oxide dissolved in cryolite.
When bauxite is treated with sodium hydroxide solution, what happens to:
- the aluminium oxide,
- the iron (III) oxide?
Name the following:
The substance added along with aluminium in the Hall-Heroult's process.
Correct the following statement :
Haematite is the chief ore of aluminium.
The sketch below illustrates the refin ing of aluminium by Hoope's process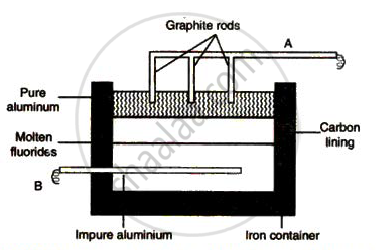
(a) Which of A and B is th e cathode and which one is the anode?
(b) What is the electroly te in the tank?
( c) What material is used for th e cathode?
The following question relate to the extraction of aluminium by electrolysis.
Explain why is it necessary to renew the anode periodically.
Describe the role played in the extraction of aluminum:
Sodium hydroxide
Name the compound added to pure alumina to lower the fusion temperature during the electrolytic reduction of alumina.
Name the alloy used for the following purpose.
Making medals
State the relevant reason for the following:
A layer of powered coke is used over the electrolytic mixture in Hall Heroult’s process.
