Advertisements
Advertisements
Question
Name the solution used to react with bauxite as a first step in obtaining pure aluminium oxide, in the Baeyer's process.
Solution
Conc. caustic soda
APPEARS IN
RELATED QUESTIONS
Both brass and bronze contain copper as major constituents. Name other elements in these alloys.
How is ore purified (give equations also)
The sketch below illustrates the refin ing of aluminium by Hoope's process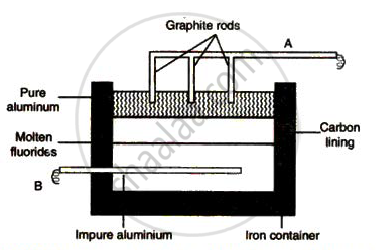
(a) Which of A and B is th e cathode and which one is the anode?
(b) What is the electroly te in the tank?
( c) What material is used for th e cathode?
Name the compound added to pure alumina to lower the fusion temperature during the electrolytic reduction of alumina.
Explain why it is preferably to use a number of graphite electrodes as anode instead of a single electrode, during the above electrolysis.
Explain with reason:
A neutral gas other than oxygen is formed at the anode during the electrolysis of fused alumina.
Name the alloy used for the following purpose.
Making parts of watches
Name a metal which is found abundantly in the Earth's crust.
State the relevant reason for the following:
Graphite anodes are continuously replaced during the electrolysis of alumina.
State true or false.
Bauxite is the main ore of aluminium.
