Advertisements
Advertisements
Question
Both brass and bronze contain copper as major constituents. Name other elements in these alloys.
Solution
- The other element in Brass is Zinc.
- The other elements in Bronze are Tin and Zinc.
APPEARS IN
RELATED QUESTIONS
Give the chemical names and formulae of the main ores of zinc.
Name three alloys of steel. Give their compositions and uses.
Aluminium is extracted from its chief ore, bauxite. The ore is first purified and then the metal is
extracted from it by electrolytic reduction
Mention one reason for the use of aluminium in thermite welding ?
Give reason for the following:
Extraction of aluminium was very difficult in the beginning.
How is aluminium obtained from pure ore?
A to F below relate to the source and extraction of either zinc or aluminium.
- Bauxite
- Coke
- Cryolite
- Froth floatation
- Sodium hydroxide solution
- Zinc blende
Fill in the blanks using the most appropriate words from A to F:
(i) The ore from which aluminium is extracted must be treated with ______ so that pure aluminium oxide can be obtained.
(ii) Pure aluminium oxide is dissolved in ______ to make a conducting solution.
Name the alloy used for the following purpose.
Making medals
The chief ore of Aluminium is ______
Write the balanced chemical equation to show the concentration of ore in Baeyer’s process.
Sodium aluminate to aluminium hydroxide
The following sketch illustrates the process of conversion of Alumina to Aluminium:
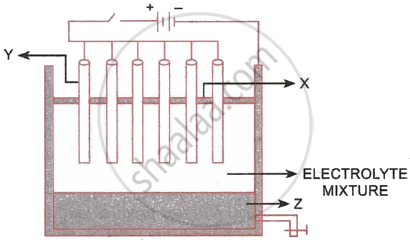
- Name the constituent of the electrolyte mixture which has a divalent metal in it.
- Name the powdered substances ‘X’ sprinkled on the surface of the electrolyte mixture.
- What is the name of the process?
- Write the reactions taking place at the electrodes ‘Y’ (anode) and ‘Z’ (cathode), respectively.
