Advertisements
Advertisements
प्रश्न
For the substance given below, describe the role played in the extraction of aluminium.
Cryolite
उत्तर
- It lowers the fusion temperature from 2050°C to 950°C and enhances conductivity.
- This, in turn, improves the electrolyte's conductivity and reduces the need for electricity, which is the primary energy source for alumina reduction.
APPEARS IN
संबंधित प्रश्न
Answer the following questions with respect to the electrolytic process in the extraction of aluminum:
Explain why powdered coke is sprinkled over the electrolytic mixture.
How is ore purified (give equations also)
The sketch below illustrates the refin ing of aluminium by Hoope's process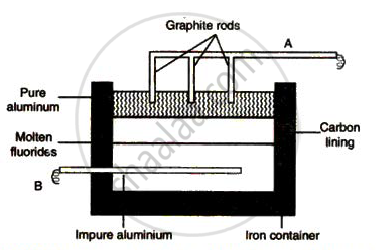
(a) Which of A and B is th e cathode and which one is the anode?
(b) What is the electroly te in the tank?
( c) What material is used for th e cathode?
The following question relate to the extraction of aluminium by electrolysis.
Give the equation for the reaction that takes place at the cathode
Describe the role played in the extraction of aluminum:
Sodium hydroxide
Name the following:
A metal which is always present in the amalgam.
Name the alloy used for the following purpose.
Making parts of watches
Name a metal which is found abundantly in the Earth's crust.
The chief ore of Aluminium is ______
The compound that is not a constituent of the electrolytic mixture used in Hall-Heroult's process is ______.
