Advertisements
Advertisements
Question
How will you convert the following in not more than two steps:
Benzoic acid to Benzaldehyde
Solution
Benzoic acid to Benzaldehyde
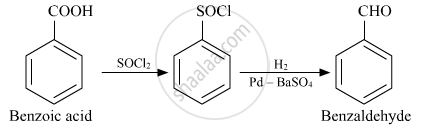
APPEARS IN
RELATED QUESTIONS
How van’t Hoff factor is related to the degree of dissociation?
Predict whether van’t Hoff factor, (i) is less than one or greater than one in the following:
CH3COOH dissolved in water
The Van't Hoff factor (i) for a dilute aqueous solution of the strong elecrolyte barium hydroxide is (NEET) ______.
We have three aqueous solutions of NaCl labelled as ‘A’, ‘B’ and ‘C’ with concentrations 0.1 M, 0.01 M and 0.001 M, respectively. The value of van’t Hoff factor for these solutions will be in the order ______.
Van’t Hoff factor i is given by the expression:
(i) i = `"Normal molar mass"/"Abnormal molar mass"`
(ii) i = `"Abnormal molar mass"/"Normal molar mass"`
(iii) i = `"Observed colligative property"/"Calculated colligative property"`
(iv) i = `"Calculated colligative property"/"Observed colligative property"`
What is the expected each water van't Hoff factor for and K4[F4(CN6)] when it completely dissociated in waters.
Maximum lowering of vapour pressure is observed in the case of ______.
Geraniol, a volatile organic compound, is a component of rose oil. The density of the vapour is 0.46 g L–1 at 257°C and 100 mm Hg. The molar mass of geraniol is ______ g mol–1. (Nearest Integer)
[Given: R = 0.082 L atm K–1 mol–1]
The degree of dissociation of Ca(NO3)2 in a dilute aqueous solution containing 7 g of the salt per 100 g of water at 100°C is 70%. If the vapour pressure of water at 100°C is 760 mm. The vapour pressure of the solution is ______ mm.
When 19.5 g of F – CH2 – COOH (Molar mass = 78 g mol−1), is dissolved in 500 g of water, the depression in freezing point is observed to be 1°C. Calculate the degree of dissociation of F – CH2 – COOH.
[Given: Kf for water = 1.86 K kg mol−1]
