Advertisements
Advertisements
Question
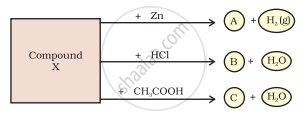
Identify the compound Y on the basis of the reactions given below. Also write the name and chemical formulae of A, B and C.
Solution
The compound ‘Y’ is sodium hydroxide. It forms A, B and C as follows :
`2"NaOH" + underset("(Y)")("Zn") -> "Na"_2"ZnO"_2 + underset("Sodium zincate (A)")("H_2(g)")`
`"NaOH" + underset("(Y)")("HCl") -> "NaCl" + underset("Sodium chloride(B)")("H"_2"O")`
`"NaOH" + underset(("Y"))("CH"_3"COOH") -> underset("Sodium acetate (Z)") ("CH"_3"COONa" + "H"_2"O")`
APPEARS IN
RELATED QUESTIONS
State the differences between acids and bases.
In terms of acidic strength, which one of the following is in the correct increasing order?
What is formed when zinc reacts with sodium hydroxide?
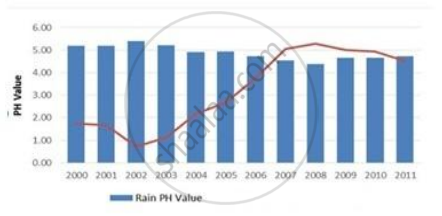
In which year is concentration of hydrogen ion the highest?
What will happen if we take dry HCl gas instead of the aqueous solution of HCl?
What will be the action of the following substances on blue litmus paper?
Dry HCl gas, Moistened NH3 gas, Lemon juice, Carbonated soft drink, Curd, Soap solution.
How would you distinguish between baking powder and washing soda by heating?
The change in colour of the moist litmus paper in the given set up is due to
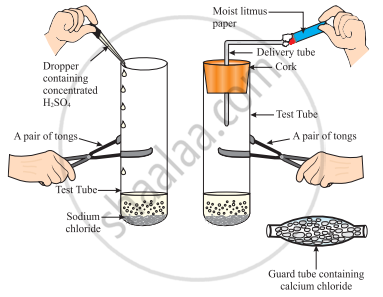
- presence of acid
- presence of base
- presence of H⁺(aq) in the solution
- presence of Litmus which acts as an indicator
Write the chemical equation for the following activity.
Dilute nitric acid was added to calcium oxide.
Write the chemical equation for the following activity.
Dilute nitric acid was added to calcium oxide.
