Advertisements
Advertisements
Question
The change in colour of the moist litmus paper in the given set up is due to
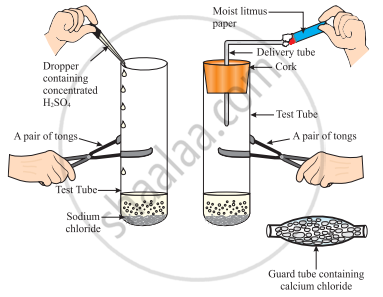
- presence of acid
- presence of base
- presence of H⁺(aq) in the solution
- presence of Litmus which acts as an indicator
Options
i and ii
Only ii
Only iii
Only iv
Solution
Only iii
APPEARS IN
RELATED QUESTIONS
When hydrogen chloride gas is prepared on a humid day, the gas is usually passed through the guard tube containing calcium chloride. The role of calcium chloride taken in the guard tube is to:
In terms of acidic strength, which one of the following is in the correct increasing order?
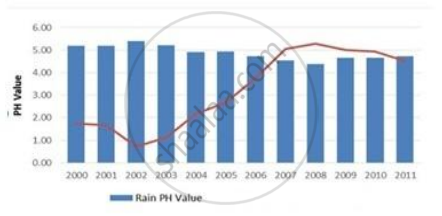
In which year is concentration of hydrogen ion the highest?
Which of the given options correctly represents the parent acid and base of Calcium Carbonate?
How would you distinguish between baking powder and washing soda by heating?
In one of the industrial processes used for the manufacture of sodium hydroxide, a gas X is formed as by product. The gas X reacts with lime water to give a compound Y which is used as a bleaching agent in chemical industry. Identify X and Y giving the chemical equation of the reactions involved.
A dry pellet of a common base B, when kept in open absorbs moisture and turns sticky. The compound is also of chloralkali process. Identify B What type of reaction occurs when B is treated with an acidic oxide ? Write a balanced chemical equation for one such solution.
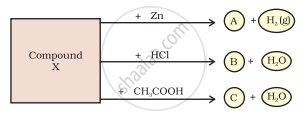
Identify the compound Y on the basis of the reactions given below. Also write the name and chemical formulae of A, B and C.
Assertion (A): It is advised that while diluting an acid one should add water to acid and not acid to water keeping the solution continuously stirred.
Reason (R): The process of dissolving an acid into water is highly exothermic.
Suggest a safe procedure of diluting a strong concentrated acid.
