Advertisements
Advertisements
Question
Identify the hydrocarbon from the given electron-dot structure:
\[\begin{array}{cc}
\phantom{}\ce{H}\phantom{....}\ce{H}\phantom{}\\
\phantom{}\ce{H}\phantom{..}\ce{\text{}^\bullet_\bullet}\phantom{..}\ce{\overset{\bullet\phantom{.}\bullet}{\underset{\bullet\phantom{.}\bullet}{C}}}\phantom{..}\ce{\text{}^\bullet_\bullet}\phantom{..}\ce{\overset{\bullet\phantom{.}\bullet}{\underset{\bullet\phantom{.}\bullet}{C}}}\phantom{..}\ce{\text{}^\bullet_\bullet}\phantom{..}\ce{H}\phantom{}\\
\phantom{}\ce{H}\phantom{....}\ce{H}\phantom{}\\
\end{array}\]
Solution
Ethane
APPEARS IN
RELATED QUESTIONS
What do you mean by catenation?
The general molecular formula of alkynes is ______
Biodegradable detergents are made of ______ chain hydrocarbons.
Answer the following question using the data given below:
- Assertion: Alkanes are saturated hydrocarbons.
- Reason: Hydrocarbons consist of covalent bonds.
Dead and decaying plants and animals release ______ gas.
______ is an odourless and highly inflammable gas.
Which gas is used as fuels and solvents in the laboratory?
Hydrocarbons are less dense than ______.
Sewage sludge can also be decomposed by microorganisms to produce methane gas along with impurities like carbon dioxide and ______.
Match the following:
| 1. | Hydrocarbons | a. | Marsh gas |
| 2. | Methane | b. | Cleanest fuel |
| 3. | Butane | c. | Catenation |
| 4. | CNG | d. | Polystyrene |
Write any 2 properties of hydrocarbons.
Identify saturated and unsaturated hydrocarbons from the given structural formula:
| \[\begin{array}{cc} \phantom{}\ce{H}\phantom{..}\ce{H}\phantom{}\\ \phantom{}|\phantom{...}|\phantom{}\\ \ce{H-C-C-H}\\ \phantom{}|\phantom{...}|\phantom{}\\ \phantom{}\ce{H}\phantom{..}\ce{H}\phantom{} \end{array}\] |
\[\begin{array}{cc} \phantom{..}\ce{H}\phantom{........}\ce{H}\phantom{.}\\ \phantom{.}\backslash\phantom{......}/\phantom{}\\ \ce{C = C}\\ \phantom{}/\phantom{......}\backslash\phantom{}\\ \phantom{}\ce{H}\phantom{........}\ce{H}\phantom{} \end{array}\] |
| (1) | (2) |
Draw electron dot structure for the following:
\[\begin{array}{cc}
\phantom{}\ce{H}\phantom{..}\ce{H}\phantom{}\\
\phantom{}|\phantom{...}|\phantom{}\\
\ce{H-C-C-H}\\
\phantom{}|\phantom{...}|\phantom{}\\
\phantom{}\ce{H}\phantom{..}\ce{H}\phantom{}
\end{array}\]
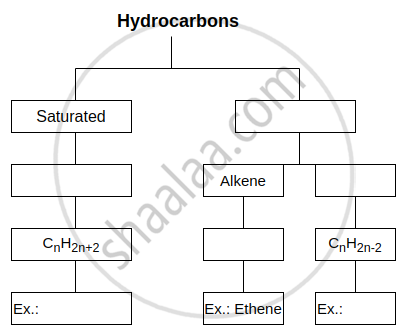
Complete the following flow chart: