Advertisements
Advertisements
Question
If you apply equal amount of heat to a solid, liquid, and gas individually, which of the following will have more expansion?
Options
Solid
Liquid
Gas
All of them
Solution
Gas
APPEARS IN
RELATED QUESTIONS
Explain the terms melting ?
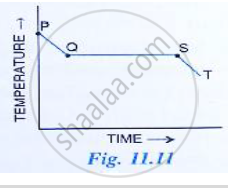
Fig 11. 11 shows the variation in temperature with time when some wax cools from the liquid phase to the solid phase.
(i) In which part of the curve, the wax is in liquid phase?
(ii) What does the part QS of the curve represent?
(iii) In which part of the curve, the wax will be the in the liquid as well as solid phase?
(iv) In which part of the curve, the wax is in solid phase?
Find the result of mixing 10 g of ice at - 10℃ with 10 g of water at 10℃. Specific heat capacity of ice = 2.1 J kg-1 K-1, Specific latent heat of ice = 336 J g-1 and specific heat capacity of water = 4.2 J kg-1 K-1.
Express -400C on the Fahrenheit scale.
Write the SI unit of:
(i) Amount of heat
(ii) Heat capacity
(iii) Specific Heat capacity
Ice cream appears colder to the mouth than water at 0°C. Give a reason.
Fill in the following blank using suitable word:
Evaporation causes ..........
200 g of hot water at 80°C is added to 300 g of cold water at 10 °C. Calculate the final temperature of the mixture of water. Consider the heat taken by the container to be negligible. [specific heat capacity of water is 4200 J kg-1 °C-1]
A calorimeter is a device used to measure the ______.
