Advertisements
Advertisements
Question
In a crystal of diamond:
(i) How many carbon atoms are present per unit cell?
(ii) What type of lattice does diamond crystallize in?
(iii) How many carbon atoms surround each carbon atom?
(iv) How are they arranged?
Solution
i)
Four carbon atoms are present per unit cell.
(ii)
Diamond crystallizes in a network type of lattice with the free arrangement of carbon atoms.
(iii)
Four carbon atoms surround each carbon atom.
(iv)
Four carbon atoms are tetrahedrally arranged around each central carbon atom.
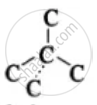
Tetrahedral arrangement
APPEARS IN
RELATED QUESTIONS
Describe triclinic crystal lattice with the help of a diagram.
Give the significance of a ‘lattice point’.
Name the parameters that characterize a unit cell.
Which of the following lattices has the highest packing efficiency (i) simple cubic (ii) body-centred cubic and (iii) hexagonal close-packed lattice?
What is meant by the term 'coordination number'?
An ionic crystal lattice has limiting value of radius ratio as 0.414 to 0.732; the co-ordination number of its cation is _______.
(A) 6
(B) 4
(C) 3
(D) 12
Calculate the number of atoms present in 2 gram of crystal which has face-centred cubic
(FCC) crystal lattice having edge length of 100 pm and density 10 gcm−3
For a crystal of sodium chloride, state:
1) The type of lattice in which it crystallises
2) The coordina~ion number of each sodium ion and chloride ion in the crystal lattice.
3) The number of sodium ions and chloride ions present in a unit cell of sodium chloride.
4) The structural arrangement of the sodium chloride crystaL
How will you obtain pure potassium permanganate (KMnO4) crystals from its ore, pyrolusite? Give the steps involved and the reactions.
The yellow colour in NaCl crystal is due to ____________.
