Advertisements
Advertisements
Question
In an accelerator experiment on high-energy collisions of electrons with positrons, a certain event is interpreted as annihilation of an electron-positron pair of total energy 10.2 BeV into two γ-rays of equal energy. What is the wavelength associated with each γ-ray? (1BeV = 109 eV)
Solution
Total energy of two γ-rays:
E = 10. 2 BeV
= 10.2 × 109 eV
= 10.2 × 109 × 1.6 × 10−10 J
Hence, the energy of each γ-ray:
`"E'" = "E"/2`
= `(10.2 xx 1.6 xx 10^(-10))/2`
= 8.16 × 10−10 J
Planck’s constant, h = 6.626 × 10−34 Js
Speed of light, c = 3 × 108 m/s
Energy is related to wavelength as:
`"E'" = "hc"/lambda`
∴ `lambda= "hc"/"E'"`
= `(6.626 xx 10^(-34) xx 3 xx 10^8)/(8.16 xx 10^(-10))`
= 2.436 × 10−16 m
Therefore, the wavelength associated with each γ-ray is 2.436 × 10−16 m.
APPEARS IN
RELATED QUESTIONS
In an experiment on the photoelectric effect, the slope of the cut-off voltage versus the frequency of incident light is found to be 4.12 × 10−15 Vs. Calculate the value of Planck’s constant.
Plot a graph showing the variation of photoelectric current with collector plate potential at a given frequency but for two different intensities I1 and I2, where I2 > I1.
Write Einstein’s photoelectric equation?
point out any two characteristic properties of photons on which Einstein’s photoelectric equation is based ?
Define the terms (i) ‘cut-off voltage’ and (ii) ‘threshold frequency’ in relation to the phenomenon of photoelectric effect.
Using Einstein’s photoelectric equation shows how the cut-off voltage and threshold frequency for a given photosensitive material can be determined with the help of a suitable plot/graph.
The frequency and intensity of a light source are doubled. Consider the following statements.
(A) The saturation photocurrent remains almost the same.
(B) The maximum kinetic energy of the photoelectrons is doubled.
A non-monochromatic light is used in an experiment on photoelectric effect. The stopping potential
A monochromatic light source of intensity 5 mW emits 8 × 1015 photons per second. This light ejects photoelectrons from a metal surface. The stopping potential for this setup is 2.0 V. Calculate the work function of the metal.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
How does one explain the emission of electrons from a photosensitive surface with the help of Einstein’s photoelectric equation?
Use Einstein's photoelectric equation to show how from this graph,
(i) Threshold frequency, and
(ii) Planck's constant can be determined.
Choose the correct answer from given options
Photons of frequency v are incident on the surface of two metals A and B of threshold frequency 3/4 v and 2/3 v, respectively. The ratio of maximum kinetic energy of electrons emitted from A to that from B is
According to Einstein's photoelectric equation, the plot of the kinetic energy of the emitted photoelectrons from a metal versus the frequency of the incident radiation gives a straight line, whose slope ______.
Each photon has the same speed but different ______.
The minimum energy required to remove an electron is called ______.
- In the explanation of photo electric effect, we assume one photon of frequency ν collides with an electron and transfers its energy. This leads to the equation for the maximum energy Emax of the emitted electron as Emax = hν – φ0 where φ0 is the work function of the metal. If an electron absorbs 2 photons (each of frequency ν) what will be the maximum energy for the emitted electron?
- Why is this fact (two photon absorption) not taken into consideration in our discussion of the stopping potential?
A student performs an experiment on photoelectric effect, using two materials A and B. A plot of Vstop vs ν is given in Figure.
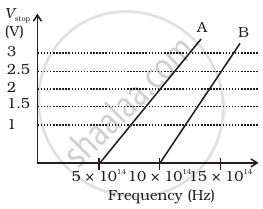
- Which material A or B has a higher work function?
- Given the electric charge of an electron = 1.6 × 10–19 C, find the value of h obtained from the experiment for both A and B.
Comment on whether it is consistent with Einstein’s theory:
Radiation of frequency 1015 Hz is incident on three photosensitive surfaces A, B and C. Following observations are recorded:
Surface A: no photoemission occurs
Surface B: photoemission occurs but the photoelectrons have zero kinetic energy.
Surface C: photo emission occurs and photoelectrons have some kinetic energy.
Using Einstein’s photo-electric equation, explain the three observations.
A photon of wavelength 663 nm is incident on a metal surface. The work function of the metal is 1.50 eV. The maximum kinetic energy of the emitted photoelectrons is ______.
