Advertisements
Advertisements
Question
In a laser tube, all the photons
Options
have same wavelength
have same energy
move in same direction
move with same speed
Solution
move with same speed
All the photons emitted in the laser move with the speed equal to the speed of light (c = 3×108 m/s).
Ideally, the light wave through the laser must be coherent, but in practical laser tubes, there is some deviation from the ideal result. Thus, the photons emitted by the laser have little variations in their wavelengths and energies as well as the directions, but the velocity of all the photons remains same.
APPEARS IN
RELATED QUESTIONS
An electron is orbiting in 5th Bohr orbit. Calculate ionisation energy for this atom, if the ground state energy is -13.6 eV.
(i) State Bohr's quantization condition for defining stationary orbits. How does the de Broglie hypothesis explain the stationary orbits?
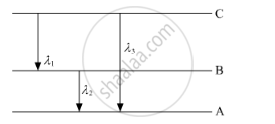
(ii) Find the relation between three wavelengths λ1, λ2 and λ3 from the energy-level diagram shown below.
What is the energy in joules, required to shift the electron of the hydrogen atom from the first Bohr orbit to the fifth Bohr orbit and what is the wavelength of the light emitted when the electron returns to the ground state? The ground state electron energy is –2.18 × 10–11 ergs.
How many electrons in an atom may have the following quantum numbers?
n = 4, `m_s = -1/2`
Using Bohr's postulates, derive the expression for the orbital period of the electron moving in the nth orbit of hydrogen atom ?
Which of the following parameters are the same for all hydrogen-like atoms and ions in their ground states?
Calculate the magnetic dipole moment corresponding to the motion of the electron in the ground state of a hydrogen atom.
Suppose in an imaginary world the angular momentum is quantized to be even integral multiples of h/2π. What is the longest possible wavelength emitted by hydrogen atoms in visible range in such a world according to Bohr's model?
According to Bohr, 'Angular momentum of an orbiting electron is quantized'. What is meant by this statement?
According to Bohr's theory, an electron can move only in those orbits for which its angular momentum is integral multiple of ____________.
What is the energy in joules released when an electron moves from n = 2 to n = 1 level in a hydrogen atom?
Which of the following is/are CORRECT according to Bohr's atomic theory?
(I) Energy is emitted when electron moves from a higher stationary state to a lower one.
(II) Orbits are arranged concentrically around the nucleus in an increasing order of energy.
(III) The energy of an electron in the orbit changes with time.
On the basis of Bohr's model, the approximate radius of Li++ ion in its ground state ifthe Bohr radius is a0 = 53 pm :
The inverse square law in electrostatics is |F| = `e^2/((4πε_0).r^2)` for the force between an electron and a proton. The `(1/r)` dependence of |F| can be understood in quantum theory as being due to the fact that the ‘particle’ of light (photon) is massless. If photons had a mass mp, force would be modified to |F| = `e^2/((4πε_0)r^2) [1/r^2 + λ/r]`, exp (– λr) where λ = mpc/h and h = `h/(2π)`. Estimate the change in the ground state energy of a H-atom if mp were 10-6 times the mass of an electron.
A hydrogen atom in its first excited state absorbs a photon of energy x × 10-2 eV and exited to a higher energy state where the potential energy of electron is -1.08 eV. The value of x is ______.
A 100 eV electron collides with a stationary helium ion (He+) in its ground state and exits to a higher level. After the collision, He+ ions emit two photons in succession with wavelengths 1085 Å and 304 Å. The energy of the electron after the collision will be ______ eV.
Given h = 6.63 × 10-34 Js.
A 20% efficient bulb emits light of wavelength 4000 Å. If the power of the bulb is 1 W, the number of photons emitted per second is ______.
[Take, h = 6.6 × 10-34 J-s]
Specify the transition of an electron in the wavelength of the line in the Bohr model of the hydrogen atom which gives rise to the spectral line of the highest wavelength ______.
The figure below is the Energy level diagram for the Hydrogen atom. Study the transitions shown and answer the following question:
- State the type of spectrum obtained.
- Name the series of spectrum obtained.

