Advertisements
Advertisements
Question
(i) State Bohr's quantization condition for defining stationary orbits. How does the de Broglie hypothesis explain the stationary orbits?
(ii) Find the relation between three wavelengths λ1, λ2 and λ3 from the energy-level diagram shown below.
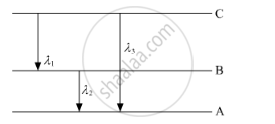
Solution
(a) Bohr's quantisation condition: According to Bohr, an electron can revolve only in certain discrete, non-radiating orbits for which total angular momentum of the revolving electron is an integral multiple of `h/(2pi)`where h is the Planck's constant.
`:.mvr=(nh)/(2pi)`
(b) Using the Rydberg formula of the spectra of hydrogen atom, we write
`1/lamda_1=R(1/n_(2^2)-1/n_(3^2)) ......(1)`
`1/lamda_2=R(1/n_(1^2)-1/n_(2^2)) ......(2)`
`1/lamda_3=R(1/n_(1^2)-1/n_(3^2)) ......(3)`
Adding (1) and (2), we find that
`1/lambda_1+1/(lambda_2)=R(1/n_(1^2)-1/n_(3^2))=1/lambda_3`
This is the required relation between the three wavelengths.
RELATED QUESTIONS
Obtain an expression for the radius of Bohr orbit for H-atom.
What is the energy in joules, required to shift the electron of the hydrogen atom from the first Bohr orbit to the fifth Bohr orbit and what is the wavelength of the light emitted when the electron returns to the ground state? The ground state electron energy is –2.18 × 10–11 ergs.
Using Bohr's postulates, derive the expression for the orbital period of the electron moving in the nth orbit of hydrogen atom ?
Write postulates of Bohr’s Theory of hydrogen atom.
If the radius of first electron orbit in hydrogen atom be r then the radius of the fourth orbit ill be ______.
The wavelength of the first time line of Ballmer series is 6563 A°. The Rydberg constant for hydrogen is about:-
In Bohr's atomic model of hydrogen, let K. P and E are the kinetic energy, potential energy and total energy of the electron respectively. Choose the correct option when the electron undergoes transitions to a higher level:
According to Bohr atom model, in which of the following transitions will the frequency be maximum?
Oxygen is 16 times heavier than hydrogen. Equal volumes of hydrogen and oxygen are mixed. The ratio of speed of sound in the mixture to that in hydrogen is ______.
State three postulates of Bohr's theory of hydrogen atom.
