Advertisements
Advertisements
Question
IUPAC name of \[\begin{array}{cc}
\phantom{....}\ce{H}\phantom{...}\ce{C4H9}\\
|\phantom{....}|\\\ce{CH3 - C - C - CH3}\\
|\phantom{....}|\\\phantom{.....}\ce{C2H5}\phantom{.}\ce{CH3}\phantom{...}\end{array}\] is
Options
3, 4, 4 – Trimethylheptane
2 – Ethyl – 3, 3, – dimethyl heptane
3, 4, 4 – Trimethyloctane
2 – Butyl – 2 – methyl – 3 ethyl – butane
Solution
3, 4, 4 – Trimethyloctane
APPEARS IN
RELATED QUESTIONS
Match the pairs.
| Column 'A' | Column 'B' | ||
| i. | Inductive effect | a. | delocalisation of π electrons |
| ii. | Hyperconjugation | b. | displacement of π electrons |
| iii. | Resonance effect | c. | delocalisation of σ electrons |
| d. | displacement of σ electrons | ||
| A covalent bond in tert-butyl bromide breaks in a suitable polar solvent to give ions. |
- Name the anion produced by this breaking of a covalent bond.
- Indicate the type of bond breaking in this case.
- Comment on the geometry of the cation formed by such bond cleavage.
In the hydrocarbon
![]() the state of hybridisation of carbon 1,2,3,4 and 7 are in the following sequence.
the state of hybridisation of carbon 1,2,3,4 and 7 are in the following sequence.
The IUPAC name of the compound\[\begin{array}{cc}\ce{CH3-CH=C-CH2-CH3}\\
|\phantom{..}\\\phantom{...............}\ce{CH2 - CH2 - CH3}\end{array}\] is
Give the IUPAC names of the following compound.
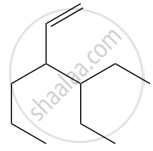
Give the IUPAC names of the following compound.
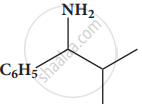
Give the structure for the following compound.
tertiary butyl iodide
Give the structure for the following compound.
2,2-dimethyl-1-chloropropane
What is the IUPAC name of the following compound?
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH2 - CH - CH2 - CH2 - CH3}\\
\phantom{}|\phantom{.....}|\phantom{.............}|\phantom{...............}\\
\phantom{}\ce{CH3}\phantom{.}\ce{CH3}\phantom{..........}\ce{C2H5}\phantom{...........}
\end{array}\]
In the trivial system which prefix will be used for the following compound?
\[\begin{array}{cc}
\ce{CH3}\phantom{.}\\
\phantom{...}\backslash\\
\ce{CH3 - C -}\\
\phantom{...}/\\
\ce{CH3}\phantom{.}\end{array}\]
