Advertisements
Advertisements
Question
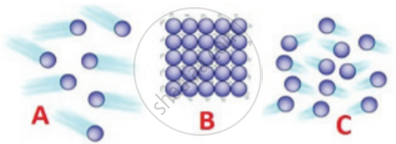
Matter in any state is composed of particles. Compare the three states of matter i,e. solids, liquids, and gases with reference to:
(a) intermolecular space
(b) the intermolecular force of attraction
(c) movement of particles
Solution
| Characteristics | Solids | Liquids | Gases |
| Intermolecular space | Minimum space | More than solids | Maximum space |
| Intermolecular force of attraction | Very strong | Less strong | Very weak |
| movement of particles | About their own position | In continuous motion | In any random direction |
RELATED QUESTIONS
Name the smallest particle from which matter is made up.
Give reasons
A gas fills up the space available to it.
Fill in the blanks:
Any matter which has a definite ______ but no definite shape is called a ______.
Write whether the following statement is true or false:
If the container in which a gas is collected has an opening, the gas will flow out and spread itself indefinitely
Describe simple experiments to prove that liquids have a definite volume
In solids, the space between the particles is less than in ______.
Identify the state of matter based on the arrangement of the molecules.
Gases have neither a definite shape nor a definite volume.
Which of the following are homogeneous in nature?
- ice
- wood
- soil
- Air
______ particles are tightly packed into a definite shape.
