Advertisements
Advertisements
प्रश्न
Matter in any state is composed of particles. Compare the three states of matter i,e. solids, liquids, and gases with reference to:
(a) intermolecular space
(b) the intermolecular force of attraction
(c) movement of particles
उत्तर
| Characteristics | Solids | Liquids | Gases |
| Intermolecular space | Minimum space | More than solids | Maximum space |
| Intermolecular force of attraction | Very strong | Less strong | Very weak |
| movement of particles | About their own position | In continuous motion | In any random direction |
संबंधित प्रश्न
Give reasons
Liquids and gases flow but solids do not?
Give reasons
When an empty gas jar is inverted over a gas jar containing a coloured gas, the gas also spreads into the empty jar
Fill in the blanks:
Any matter which has a definite ______ but no definite shape is called a ______.
Describe simple experiments to prove that solids have a definite volume
Describe simple experiments to prove that liquids have no definite shape
State the property of the following substance.
Manganese dioxide, a catalyst which alters the rate of a chemical reaction is black in colour.
See the given pictures and write down the state of each object. (Solid, Liquid, Gas)
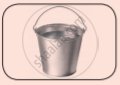 |
 |
 |
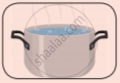 |
 |
| _____ | ______ | ______ | ______ | ______ |
 |
 |
 |
 |
 |
| ______ | _______ | ______ | ______ | ______ |
Which of the following is a solid?
The force of attraction between liquid particles is more than solid particles.
Gases have neither a definite shape nor a definite volume.
