Advertisements
Advertisements
प्रश्न
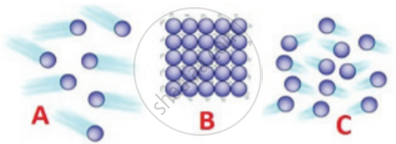
Matter in any state is composed of particles. Compare the three states of matter i,e. solids, liquids, and gases with reference to:
(a) intermolecular space
(b) the intermolecular force of attraction
(c) movement of particles
उत्तर
| Characteristics | Solids | Liquids | Gases |
| Intermolecular space | Minimum space | More than solids | Maximum space |
| Intermolecular force of attraction | Very strong | Less strong | Very weak |
| movement of particles | About their own position | In continuous motion | In any random direction |
संबंधित प्रश्न
Compare the properties of solids, liquids and gases in tabular form.
Give reasons
When an empty gas jar is inverted over a gas jar containing a coloured gas, the gas also spreads into the empty jar
The three main states of matter are solids, liquids, and gases. Compare the three states with reference to the following characteristics of matter
(a) volume
(b) shape
(c) compressibility
(d) diffusion.
Write the states of water.
Which of the following are the states of matter?
Which of the following is a solid?
Complete the given analogy.
Large Inter-particle space: Gas:: ______ :solid.
A rubber ball changes its shape on pressing. Can it be called a solid?
Identify the state of matter based on the arrangement of the molecules.
What is the physical state of water at 100ºC?
