Advertisements
Advertisements
Questions
Name a non-metallic element which is a conductor of electricity.
Name the following:
The non-metallic substance which is a good conductor of electricity.
Solution
Carbon (graphite) is a non-metallic element that is an electricity conductor.
RELATED QUESTIONS
Differentiate between the terms strong electrolyte and weak electrolyte. (stating any two differences)
The electrolysis of acidified water is an example of :
(A) Reduction
(B) Oxidation
(C) Redox reaction
(D) Synthesis
What particles are present in pure lead bromide?
The following is an extract from metals in the service of man, Alexander and street /Pelican 1976': Alumina (aluminium oxide) has a very high melting point over 2000oC, so that I cannot readily be liquefied. However, conversion of alumina to aluminium and oxygen, by electrolysis, an occur when it is dissolved in some other substance.
(a) Which solution is used to react with bauxite as first step in obtaining pure aluminium oxide?
(b) The aluminium oxide for the electrolytic extraction of aluminium is obtained by heating aluminium hydroxide. Write the equation for this reaction.
(c) Name the element which serves both as the anode and the cathode in the extraction of aluminium.
(d) Write the equation for the reaction that occurs at the cathode during the extraction of aluminium.
(e) Write the equation for the reaction that occurs at the cathode during extraction of aluminium by electrolysis.
(f) Give the equation for the reaction that occurs at the anode when aluminium is purified by electrolysis.
Copper sulphate solution is electrolyzed using copper electrodes. Study the diagram given alongside and answer the questions that follow.
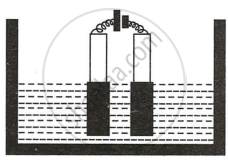
- Which electrode to your left or right is known as the oxidizing electrode and why?
- Write the equation representing the reaction that occurs.
- State two appropriate observations for the above electrolysis reactions.
Write the equation taking place at the anode.
Give reasons why although copper is a good conductor of electricity, it is a non-electrolyte.
Select the ion, that would get selectively discharge from the aqueous mixture of the ions listed below :
\[\ce{SO^{2-}_{4}}\], \[\ce{NO^{-}_{3}}\], \[\ce{OH-}\]
Classify the following substance:
Ammonium hydroxide
Classify the following substance:
Dilute sulphuric acid
