Advertisements
Advertisements
Question
Name the reagents used to convert phenol into
- picric acid
- p-benzoquinone
Solution
- Concentrated nitric acid in the presence of conc. H2SO4 is used to convert phenol into picric acid.
- Chromic anhydride or sodium dichromate in the presence of H2SO4 is used to convert phenol into p-benzoquinone.
APPEARS IN
RELATED QUESTIONS
Answer in one sentence/ word.
Hydroboration-oxidation of propene gives _________.
Answer in one sentence/ word.
Write the name of the electrophile used in Kolbe’s Reaction.
Answer in brief.
Give the reagents and conditions necessary to prepare phenol from Benzene sulfonic acid.
When vapours of tert.butyl alcohol are passed over hot copper, it gives _____________
The reagents used to convert phenol to 2,4,6-tribromophenol is _____________
Arrange the following in decreasing order of acid strength.
CH3OH, CH3–CH2–OH, CH3–CH(OH)–CH3, (CH3)3–C–OH
Write the reaction to get aspirin from salicylic acid.
Write Kolbe’s reaction.
Write chemical equation of acetyl chloride with phenol
An unknown alcohol is treated with Lucas reagent. Explain how you will determine whether the alcohol is primary, secondary or tertiary. Indicate by chemical equation the reaction between isopropyl alcohol and Lucas reagent.
How will you bring about the following conversions?
2-methyl propan-2-ol to 2-methylpropene
An organic compound gives hydrogen on reaction with sodium metal. It forms an aldehyde with molecular formula C2H4O on oxidation with pyridinium chlorochromate. Give the chemical equations in support of these observations.
Number of oxygen atoms present in salicylaldehyde are ______.
α-butylene when subjected to hydroboration oxidation reaction, yields ______.
The CORRECT decreasing order of boiling points for isomeric primary (1°), secondary (2°) and tertiary (3°) alcohols is ____________.
Phenol is ____________.
Which of following elements does not react with hot concentrated sulphuric acid?
Cumene is used in the commercial method for the manufacture of ____________.
The most resistant alcohol towards oxidation reaction is:
Identify the product B in following conversion.
\[\ce{Chlorobenzene + H2O ->[Cu, 673 K][Pressure] A ->[conc. H2SO4][373 K] B}\]
Name the catalyst used in commercial method of preparation of phenol.

Product (B) in this reaction is:
Which of the following compounds has lowest boiling point?
Which of the following compounds reacts immediately with Lucas reagent?
The major product obtained in the following reaction is

Arrange the following compounds in an increasing order of their solubility in water:
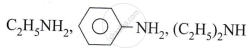
Convert the following :
cumene to phenol.
