Advertisements
Advertisements
Question
An unknown alcohol is treated with Lucas reagent. Explain how you will determine whether the alcohol is primary, secondary or tertiary. Indicate by chemical equation the reaction between isopropyl alcohol and Lucas reagent.
Solution
Primary, secondary and tertiary alcohols can be distinguished from each other in the laboratory using Lucas reagent (conc. HCl and ZnCl2).
- An unknown alcohol is treated with Lucas reagent.
The reaction involved is: \[\ce{R - OH ->[HCl][ZnCl2] R - Cl}\] - Depending on the observation an unknown alcohol can be identified as primary, secondary or tertiary.
Test Observation Inference Unknown Alcohol + Lucas reagent Reagent turns turbid on heating. Primary alcohol is present. Unknown Alcohol + Lucas reagent Reagent turns turbid slowly, without heating. Secondary alcohol is present. Unknown Alcohol + Lucas reagent Reagent turns turbid immediately, without heating. Tertiary alcohol is present. - The reaction between isopropyl alcohol and Lucas reagent can be given as:
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 + HCl + ZnCl2 -> CH3 - CH - CH3}\\
\phantom{..}|\phantom{......................................}|\phantom{..}\\
\phantom{.....}\ce{\underset{\text{Isopropyl alcohol (2° alcohol)}}{OH} \phantom{....................}\underset{\text{Isopropyl chloride}}{Cl}}\phantom{........}
\end{array}\]
The reagent will turns turbid slowly, without heating.
APPEARS IN
RELATED QUESTIONS
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
When vapours of tert.butyl alcohol are passed over hot copper, it gives _____________
The reagents used to convert phenol to 2,4,6-tribromophenol is _____________
Write IUPAC name of crotonyl alcohol.
Write the reaction to get aspirin from salicylic acid.
How will you bring about the following conversions?
acetone to 2-methylpropan-2-ol
Which of the following compounds does not react with bromine in alkaline medium?
The CORRECT decreasing order of boiling points for isomeric primary (1°), secondary (2°) and tertiary (3°) alcohols is ____________.
The secondary alcohol is ____________.
Phenol is ____________.
Carbolic acid is oxidised by acidified sodium dichromate to give ______.
In Dow process, chlorobenzene is converted into phenol by ____________.
Which of the following is INCORRECT regarding phenol?
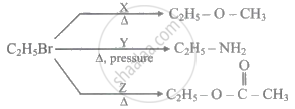
Identify reagents X, Y and Z.
Which of the following conversion explains the acidic nature of alcohols?
Which following reagent is used to obtain alkene from alcohol?
Which among the following compounds is used to prepare solid fuel using calcium acetate?
Identify 'Z' in the following series of reaction:
\[\ce{Butan - 2 - ol ->[PCl3] X ->[alco. KOH] Y ->[i) H2SO4][ii) H-OH/heat] Z}\]
Identify the product obtained when phenol is treated with bromine water?

Product (B) in this reaction is:
The major product obtained in the following reaction is

What is the action of nitrous acid on aniline?
Arrange the following compounds in an increasing order of their solubility in water:
Write the chemical reaction when hot copper is treated with Vapours of 1° (primary) alcohol.
Write the chemical reaction when hot copper is treated with Vapours of 2° (secondary) alcohol.
